2026-03-19 東北大学
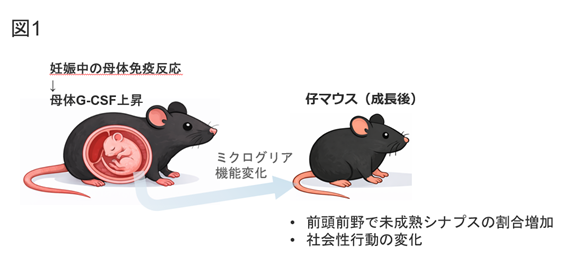
図1. 妊娠期と仔の脳発達への影響(本研究の概念図)
妊娠中の免疫反応により免疫因子G-CSFが上昇すると、仔の前頭前野で未成熟シナプス構造の割合が増加し、成長後の社会性行動にも変化が生じることがマウス研究で示されました。
<関連情報>
- https://www.tohoku.ac.jp/japanese/2026/03/press20260319-02-G-CSF.html
- https://www.tohoku.ac.jp/japanese/newimg/pressimg/tohokuuniv-press20260319_web02_G-CSF.pdf
- https://www.sciencedirect.com/science/article/pii/S0889159126002825?via%3Dihub
母体顆粒球コロニー刺激因子は、子孫のシナプス成熟と社会的行動を変化させる Maternal granulocyte colony-stimulating factor alters synaptic maturation and social behaviors in offspring
Hinako Kirikae, Karina Kimura, Jinghang Fu, Zhengkang Sun, Hongbo Wang, Haruka Shibuya, Yoshiyuki Kasahara, Hirofumi Miyazaki, Yui Yamamoto, Mai Sakai, Zhiqian Yu, Shohei Ochi, Fumito Naganuma, Takeo Yoshikawa, Takashi Namba, Noriko Osumi, Hiroaki Tomita, Yuji Owada, Motoko Maekawa
Brain, Behavior, and Immunity Available online: 11 March 2026
DOI:https://doi.org/10.1016/j.bbi.2026.106534
Highlights
- Maternal immune activation (MIA) elevates G-CSF in both maternal and fetal compartments.
- Prenatal G-CSF exposure altered brain structure, behavior, and gene expression in both sexes.
- G-CSF disrupts synapse maturation and modulates microglial activity in vitro.
- G-CSF functions as a non-inflammatory mediator linking MIA to neurodevelopmental risk.
Abstract
Neurodevelopmental disorders, including autism spectrum disorder (ASD), arise from complex interactions between genetic and environmental factors. Maternal immune activation (MIA) is a key environmental risk factor that disrupts embryonic neurodevelopment, primarily through inflammatory cytokines. However, the contribution of non-inflammatory cytokines, particularly hematopoietic growth factors, remains poorly understood. Here, we identified granulocyte colony-stimulating factor (G-CSF) as a candidate mediator of MIA-induced neurodevelopmental alterations. Polyinosinic:polycytidylic acid [poly(I:C)] administration to pregnant dams at embryonic day 12.5 (E12.5) significantly increased G-CSF levels in both maternal plasma and embryonic tissue. To assess its contribution to neurodevelopmental alterations, we administered human G-CSF (hG-CSF) to pregnant dams at E12.5. At the structural level, male offspring exposed to prenatal hG-CSF showed increased dendritic spine density and a higher proportion of immature spines in the medial prefrontal cortex. Behaviorally, both male and female offspring exhibited altered social preference. Bulk RNA-seq analysis of the prefrontal cortex revealed altered enrichment of pathways related to synapse organization, translation, and mitochondrial function in both sexes, with opposite directions of enrichment in males and females. In vitro, G-CSF attenuated synapse maturation and enhanced microglial phagocytic activity. These findings suggest that G-CSF may contribute to MIA-associated neurodevelopmental alterations, potentially through disrupted synapse maturation and microglial function. Our results highlight a hematopoietic pathway that may contribute to mechanisms underlying neurodevelopmental disorders, including ASD.


