2026-04-03 熊本大学
<関連情報>
- https://www.kumamoto-u.ac.jp/whatsnew/seimei/20260403-2
- https://www.kumamoto-u.ac.jp/daigakujouhou/kouhou/pressrelease/wgt3jw/release260403-2.pdf
- https://ashpublications.org/bloodadvances/article/doi/10.1182/bloodadvances.2025018710/567540/IL-6-driven-POU2AF1-and-ELL2-are-key-regulators-of
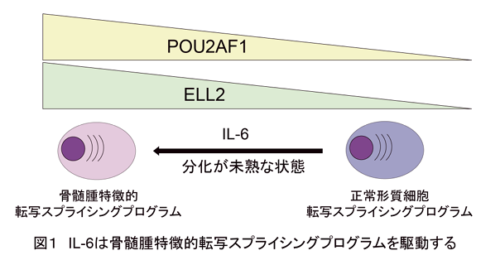
IL-6によって誘導されるPOU2AF1とELL2は、多発性骨髄腫特有の転写およびスプライシングプログラムの主要な制御因子である IL-6-driven POU2AF1 and ELL2 are key regulators of multiple myeloma-distinct transcriptional and splicing programs
Yasuyo Ohguchi,Masahiko Ajiro,Daisuke Ogiya,Takeshi Masuda,Yawara Kawano,Shingo Usuki,Tomoaki Koga,Shinjiro Hino, Dr.,Takeshi Harada,Satoru Takahashi,Seiji Okada,Jun-ichirou Yasunaga,Goro Sashida,Sumio Ohtsuki,Mitsuyoshi Nakao,Takashi Minami,Akihide Yoshimi,Hiroto Ohguchi
Blood Advances Published:April 2, 2026
DOI:https://doi.org/10.1182/bloodadvances.2025018710
Key Points
- IL-6-driven POU2AF1 and ELL2 confer the robustness of MM-distinct transcriptional program.
- POU2AF1 is also engaged in the organization of nuclear speckles and IL6-mediated alternative splicing.
Multiple myeloma (MM) is a plasma cell neoplasm that depends on the bone marrow (BM) microenvironment; however, the underlying mechanisms of epigenetic contribution to the pathogenesis of MM are incompletely understood. Here, we delineate the epigenetic-driven transcriptional and splicing regulation crucial for MM. We recharacterized transcriptional program induced by IL-6/JAK/STAT3 pathway by integrating ChIP-seq, transcriptomic analyses, and CRISPR knockout screening results, and identified B cell lineage factors, POU2AF1 and ELL2, as crucial IL-6/JAK/STAT3 targets essential for MM cell growth and survival. Genetic depletion of these factors significantly suppressed MM cell growth in vitro and in the xenograft model of IL-6 humanized mice. Mechanistically, POU2AF1 and ELL2 form an autoregulatory loop with IRF4 and establish MM-distinct transcriptional program representing immaturity, and IL-6/JAK/STAT3 pathway augments this program through upregulating and recruiting these factors to the MM-signature genes. Furthermore, POU2AF1 and ELL2 are essential in the regulation of IL-6-dependent alternative RNA splicing. Immunocytochemical and proteomic analyses revealed that POU2AF1 colocalizes and facilitates formation of nuclear speckles, where it interacts with multiple trans-acting splicing factors required for MM cell growth. These findings suggest the dual roles of POU2AF1 and ELL2 in coordinating transcription and RNA splicing to generate MM-associated mRNA isoforms. Finally, we showed that gapmer antisense oligonucleotides targeting POU2AF1 inhibit its expression and MM cell growth in the presence of soluble BM stromal cell factors, including IL-6. Our data demonstrate that IL-6-driven B cell-lineage factors are the vulnerability of MM cells and may represent novel therapeutic targets for this incurable tumor.