2026-05-01 京都府立大学
概念図:本研究で発見した作用とその作用機序
<関連情報>
- https://www.kpu.ac.jp/news/kenkyuuseika20260501/
- https://www.kpu.ac.jp/media/20260501Pressrelease.pdf
- https://diabetesjournals.org/diabetes/article-abstract/doi/10.2337/db25-1134/164779/Gut-Derived-GLP-1-Released-by-Rare-Sugar-d?redirectedFrom=fulltext
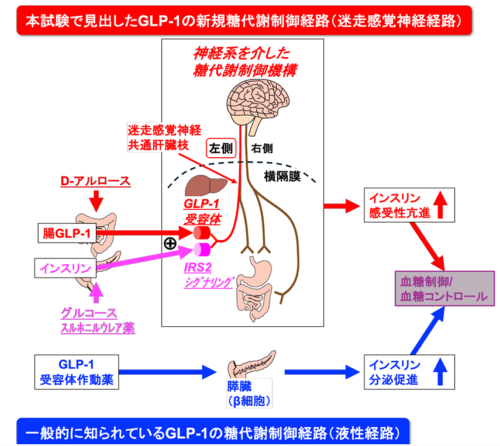
希少糖 D-アルロースによる腸由来 GLP-1 放出は、インスリンと協働して左側の迷走感覚神経を活性化することで、インスリン感受性を増強する Gut-Derived GLP-1 Released by Rare Sugar d-Allulose Cooperates With Insulin to Activate Left-Sided Vagal Afferents and Enhance Insulin Sensitivity
Kento Ohbayashi;Mamoru Tanida;Chikara Abe;Hirotaka Ishihara;Wataru Omi;Naoto Kubota;Daniel J. Drucker;Toshihiko Yada;Yusaku Iwasaki
Diabetes Published:May 01 2026
DOI:https://doi.org/10.2337/db25-1134
Glucagon-like peptide 1 receptor agonists (GLP-1RAs) ameliorate hyperglycemia by directly stimulating insulin secretion from the pancreas. In contrast, the physiological role of short-lived endogenous GLP-1 remains unclear largely because of its limited access to pancreatic β-cells. Here, we used d-allulose, a nonmetabolizable noncaloric rare sugar, as a GLP-1 secretagogue. We show that d-allulose–induced intestinal GLP-1 secretion (AIGS) cooperates with insulin to reduce blood glucose levels by enhancing insulin action, rather than insulin secretion, in male mice. This cooperation and remote signaling require left-sided vagal afferents forming the common hepatic branch, but not right-sided afferents. AIGS-enhanced insulin action required both GLP-1Rs and insulin receptor substrate 2 in these neurons. Remarkably, in db/db mice exhibiting severe insulin resistance, AIGS improved insulin resistance and hyperglycemia more rapidly and potently than the GLP-1RA exendin-4. These findings reveal that a subclass of vagal afferent neurons synergistically activated by endogenous intestinal GLP-1 and insulin does not stimulate insulin secretion but augments insulin action to improve glucose tolerance. This novel extrapancreatic GLP-1 action mediated by vagal afferents provides a promising basis for innovative type 2 diabetes therapies.
Article Highlights
- Compared with glucagon-like peptide 1 (GLP-1) receptor agonists, the physiological roles and mechanisms of endogenous, short-lived GLP-1 in glucose metabolism remain poorly understood.
- We used the rare sugar d-allulose, a noncaloric GLP-1 secretagogue, as a tool to elucidate the physiological actions of endogenous GLP-1.
- d-allulose–induced intestinal GLP-1 release cooperates with insulin to activate left-side vagal afferents, enhancing insulin action rather than insulin secretion and thereby regulating glycemic control.
- Because this acute mechanism improved hyperglycemia in type 2 diabetes to an extent comparable to that observed with GLP-1 receptor agonists, targeting GLP-1/insulin-vagal signaling may inform novel therapies and dietary or nutritional interventions for type 2 diabetes.