2026-05-04 カロリンスカ研究所(KI)
<関連情報>
- https://news.ki.se/new-treatment-with-fewer-side-effects-reduces-breast-density
- https://academic.oup.com/jnci/advance-article/doi/10.1093/jnci/djag087/8665489
乳房密度低減のためのエンドキシフェン ― KARISMAエンドキシフェン試験の結果 Endoxifen for mammographic density reduction—results from the KARISMA endoxifen trial
Per Hall, MD, PhD;Mattias Hammarström, Msc;Jenny Bergqvist, MD, PhD;Kamila Czene, PhD;Mikael Eriksson, PhD;Marike Gabrielson, PhD;José Tapia, MS;Steven Quay, MD, PhD;Stephen Nash, Msc;Magnus Bäcklund, MD, PhD
Journal of the National Cancer Institute Published:27 April 2026
DOI:https://doi.org/10.1093/jnci/djag087
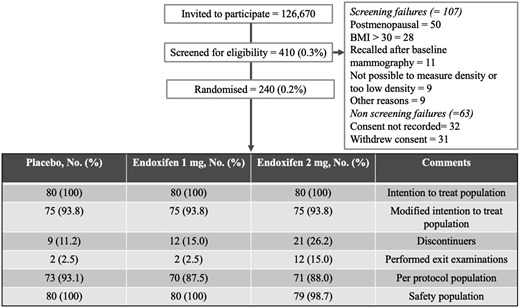
Figure 1.CONSORT diagram for the KARISMA Endoxifen trial.
Abstract
Background
(Z)-endoxifen is the tamoxifen metabolite that possesses the highest affinity to the estrogen receptor and is evolving as an alternative to tamoxifen. Mammographic breast density (MBD) change has been shown to be a proxy for tamoxifen therapy response. The objective was to measure the effect of 2 different doses of (Z)-endoxifen on MBD, safety, and side effects in healthy women.
Methods
Healthy premenopausal women included in the national Swedish screening program in Stockholm were invited to KARISMA Endoxifen, a proof of principle, dose determining, double-blinded, randomized, placebo-controlled trial. Women were randomly assigned to placebo or 1 or 2 mg of (Z)-endoxifen daily for 6 months.
Results
In all, 240 women were randomly assigned. There was a significant relative change in MBD in both (Z)-endoxifen arms compared to placebo: −19.3% (95% confidence interval [CI] = −6.15% to −32.4%) in the 1 mg arm and −26.5% (95% CI = −14.1% to −38.9%) in the 2 mg arm. The number of participants discontinuing because of adverse events related to the investigational medicinal product was 4 (placebo), 5 (1 mg), and 11 (2 mg), respectively. Participants on 2 mg of (Z)-endoxifen reported significantly higher scores of vasomotor symptoms, compared with placebo. No clinically significant changes in hematological safety tests or vital signs were noted.
Conclusion
Both 1 and 2 mg of (Z)-endoxifen significantly reduced MBD to a degree comparable to the established 20 mg dose of tamoxifen. The 1 mg dosage of (Z)-endoxifen indicated superior tolerability. Future studies are necessary to confirm impact on breast cancer incidence.
Trial Registration
ClinicalTrials.gov ID: NCT05068388.

