2026-05-13 順天堂大学
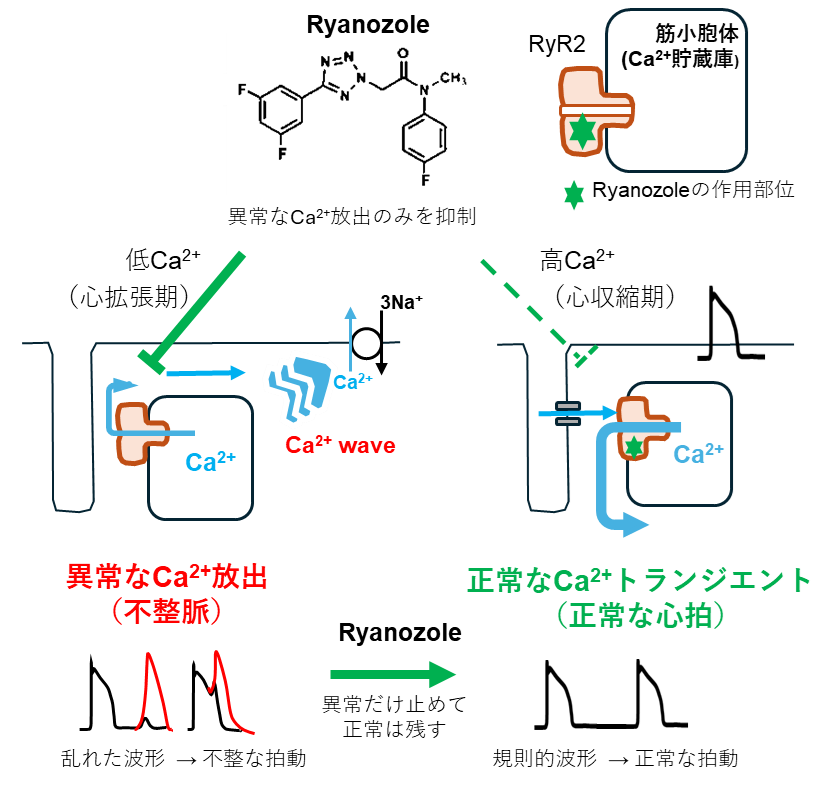
図1:本研究で明らかになったRyanozoleの作用機序
Ryanozoleは、心拡張期に不整脈の原因となる異常なCa2+放出のみを抑制し、収縮期には正常なCa2+ transientを保つことで、心機能を損なわずに不整脈を抑制する。
<関連情報>
- https://www.juntendo.ac.jp/news/26833.html
- https://bpspubs.onlinelibrary.wiley.com/doi/10.1111/bph.70459
新規RyR2選択的抑制薬は心機能を損なわずにストレス誘発性不整脈を抑制する A novel selective stabilizer of the ryanodine receptor 2 prevents stress-induced ventricular arrhythmias without impairing cardiac function
Nagomi Kurebayashi, Masami Kodama, Hana Inoue, Masato Konishi, Masami Sugihara, Takashi Murayama, Ryosuke Ishida, Koichiro Ishii, Shuichi Mori, Yukari Endo, Xi Zeng, …
British Journal of Pharmacology Published: 27 April 2026
DOI:https://doi.org/10.1111/bph.70459
Abstract
Background and Purpose
Aberrant activation of type 2 ryanodine receptors (RyR2) causes lethal arrhythmias, such as catecholaminergic polymorphic ventricular tachycardia (CPVT). Developing drugs that suppress RyR2 hyperactivation may be key to novel arrhythmia treatments. This study evaluated the antiarrhythmic potential of Ryanozole, a recently developed novel RyR2 modulator with high affinity and selectivity, using CPVT mouse models harbouring mutant RyR2s.
Experimental Approach
In vitro effects of Ryanozole were evaluated by endoplasmic reticulum (ER) Ca2+-based assay and [3H]ryanodine-binding assay using RyR2-expressing HEK293 cells. Two lines of mice with different arrhythmia severities, RyR2-R420W and RyR2-K4750Q, were employed for in vivo and ex vivo assessments. Intracellular Ca2+ signals were analysed in isolated cardiomyocytes using Cal-520. Antiarrhythmic effects were evaluated by electrocardiography (ECG) under catecholaminergic challenge in anaesthetized mice and during spontaneous arrhythmias in conscious mice. ECG and echocardiographic parameters were evaluated before and after drug administration.
Key Results
Ryanozole inhibited both wild-type and mutant RyR2s with an IC50 of 15–40 nM. Inhibition was more potent at lower cytosolic Ca2+ concentrations. Ryanozole suppressed Ca2+ waves and Ca2+ sparks without affecting action potential-evoked Ca2+ transients. Ryanozole effectively prevented adrenaline-induced arrhythmias and rapidly terminated ongoing spontaneous arrhythmias during daily activity. Importantly, Ryanozole did not impair cardiac conduction or contractility, unlike conventional antiarrhythmic drugs.
Conclusion and Implications
Ryanozole preferentially suppresses RyR2 under diastolic Ca2+ conditions, thereby preventing the arrhythmogenic trigger of aberrant Ca2+ release. This mechanism may provide potent antiarrhythmic effects while preserving cardiac function. Ryanozole is a potential therapeutic candidate for RyR2-mediated arrhythmias, such as CPVT.