2026-03-23 中国科学院(CAS)
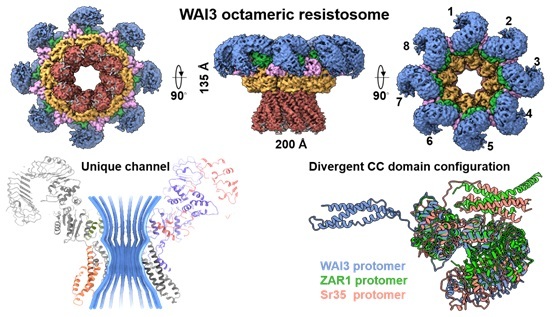
An activated wheat CCG10-NLR immune receptor forms an octameric resistosome. (Image by IGDB)
<関連情報>
- https://english.cas.cn/newsroom/research-news/202603/t20260317_1152821.shtml
- https://www.cell.com/cell/fulltext/S0092-8674(26)00231-X
活性化されたコムギCC G10 -NLR免疫受容体は八量体レジストソームを形成する An activated wheat CCG10-NLR immune receptor forms an octameric resistosome
Guanghao Guo ∙ He Zhao, ∙ Kaihong Bai ∙ … ∙ Zhiyong Liu ∙ Muniyandi Selvaraj ∙ Jonathan D.G. Jones
Cell Published:March 20, 2026
DOI:https://doi.org/10.1016/j.cell.2026.02.024
Highlights
- CCG10-NLRs WAI3 and RPS2 form octameric resistosomes upon activation
- The WAI3 resistosome triggers calcium influx and immune responses in plants
- The activation of WAI3 requires plant-specific factor(s) to trigger calcium influx
- The WAI3-like configuration may be shared by plant NLRs lacking the EDVID motif
Summary
Nucleotide-binding, leucine-rich repeat (NLR) receptors are widespread intracellular immune sensors across kingdoms. Plant G10-type coiled-coil (CCG10)-NLRs constitute a distinct phylogenetic clade that remains poorly characterized. Here, we identified a gain-of-function mutant of wheat autoimmunity 3 (WAI3GOF), which encodes a constitutively active CCG10-NLR resulting from a residue substitution in the leucine-rich repeat (LRR) domain. Cryo-electron microscopy (cryo-EM) analysis reveals that activated WAI3 assembles into a distinctive octameric resistosome. Arabidopsis RPS2, another CCG10-NLR, also forms an octamer, indicating a conserved structural property across monocot and dicot plants. The WAI3 resistosome induces a prolonged and sustained increase in cytosolic calcium, likely facilitated by a unique channel architecture arising from its divergent coiled-coil (CC) domain configuration. Notably, this domain arrangement may be shared by plant NLRs that lack the conserved EDVID (Glu-Asp-Val-Ile-Asp) motif in their CC domains. Together, our findings uncover a conserved yet previously uncharacterized NLR resistosome structure and provide insights into the plant immune receptor plasticity.