2026-05-15 京都大学iPS細胞研究所
本研究の概要
<関連情報>
- https://www.cira.kyoto-u.ac.jp/j/pressrelease/news/260515-000000.html
- https://www.cell.com/stem-cell-reports/fulltext/S2213-6711(26)00134-7
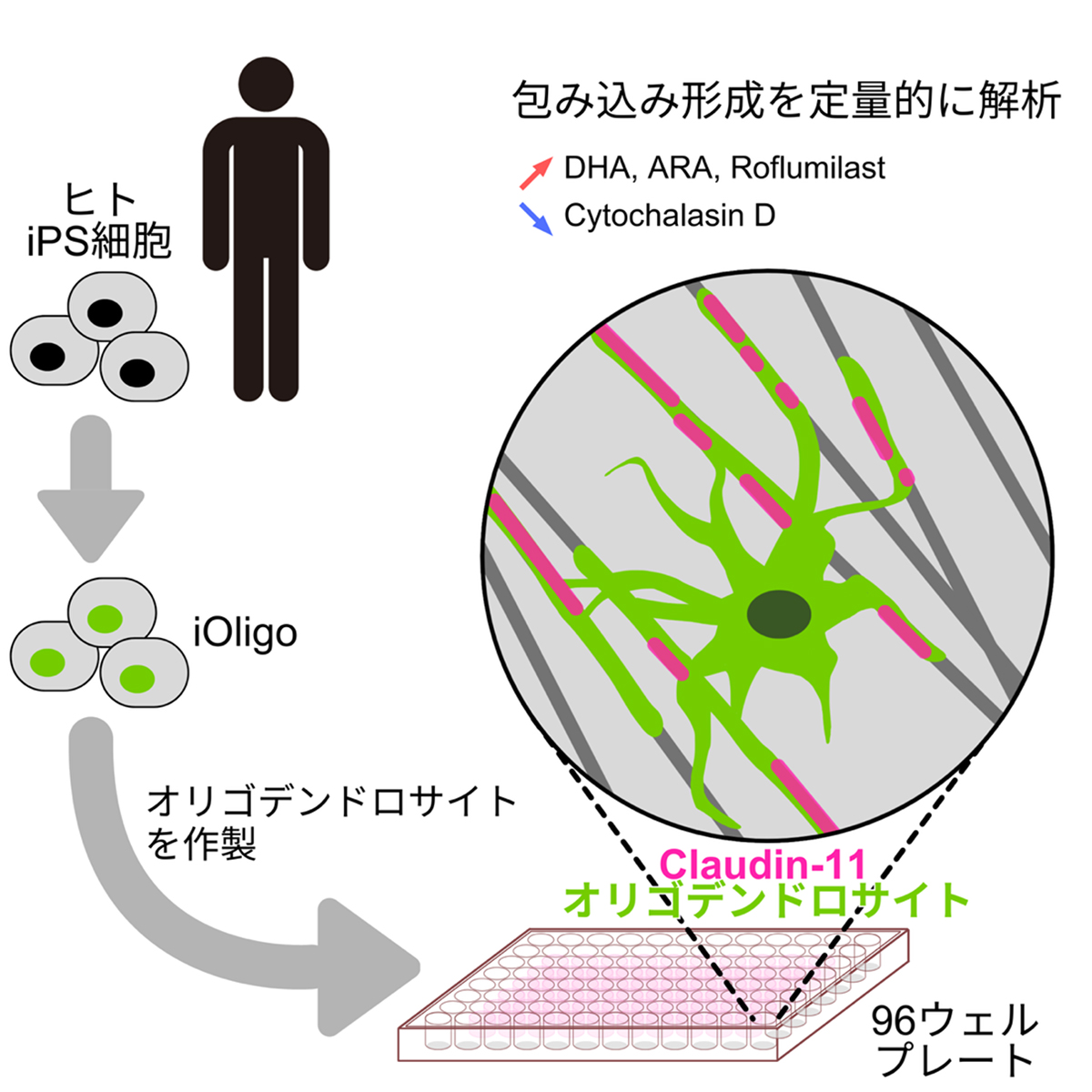
薬理学的摂動によるヒトオリゴデンドロサイト被覆の定量的分析のためのナノファイバーベースプラットフォーム Nanofiber-based platform for quantitative analysis of human oligodendrocyte ensheathment with pharmacological perturbations
Satoshi Morita ∙ Takayuki Kondo ∙ Keiko Imamura ∙ … ∙ Takayuki Izumo ∙ Yoshihiro Nakao ∙ Haruhisa Inoue
Stem Cell Reports Published:May 14, 2026
DOI:https://doi.org/10.1016/j.stemcr.2026.102923
Highlights
- Efficient differentiation of human oligodendrocytes from iPSCs within a short time
- Nanofiber scaffolds induce transcriptional priming and ensheathment behavior
- Oriented CLDN11 expression quantifies structural organization during ensheathment
- A versatile platform to study oligodendrocyte-related disorders
Summary
Oligodendrocytes are glial cells responsible for myelination in the central nervous system, and their dysfunction underlies various neurological disorders. However, existing human oligodendrocyte models are limited by low efficiency and insufficient standardization. Here, we developed a well-defined system for differentiating human oligodendrocytes from induced pluripotent stem cells using transient transcription factor expression. To recapitulate key structural aspects of oligodendrocyte-axon interactions, we cultured oligodendrocytes on nanofibers mimicking axonal topology. On this platform, ultrastructural, live-imaging, and transcriptomic analyses demonstrated dynamic oligodendrocyte-nanofiber interactions and ensheathment-like wrapping. This process was accompanied by aligned CLDN11 expression along nanofibers, providing a quantifiable structural readout without overt maturation. To validate the utility of the platform, we quantified the effects of white matter toxins and pro-ensheathment lipids. Our findings establish a robust system for evaluating oligodendrocyte ensheathment modulators, particularly those affecting cytoskeletal organization and initial sheath formation, and for investigating the pathophysiology of human oligodendrocyte-related disorders.