2026-03-09 浙江大学(ZJU)
STING restricts the replication of avian site-carrying viruses
<関連情報>
- https://www.zju.edu.cn/english/2026/0309/c19573a3138494/page.psp
- https://www.science.org/doi/10.1126/science.ads4405
STING-NF-κBシグナル伝達がインフルエンザのスピルオーバーバリアを構築する STING–NF-κB signaling builds an influenza spillover barrier
Runxin Ye, Songdi Wang, Ying Hu, Yiran Pan, […] , and Xiao-Fang Yu
Science Published:26 Feb 2026
Editor’s summary
Proteins within lung epithelial cells can block the ability of viruses to replicate. Ye et al. found that activation of STING, an important molecule in the innate immune system, could limit influenza infection by activating a transcription factor complex called NF-κB (see the Perspective by Yan). A protein encoded by the human influenza A virus, matrix protein 1 (M1), bound to STING and blocked NF-κB activation, thus helping to facilitate viral replication in infected cells. By contrast, M1 proteins from bird influenza viruses were unable to bind to STING, and their replication was inhibited. This suggests that STING and NF-κB form a molecular barrier that may help to prevent viruses that typically infect other species from replicating in human cells. —Sarah H. Ross
Structured Abstract
INTRODUCTION
Influenza A viruses (IAVs) of avian origin pose a continuous pandemic threat. For transmission to occur, avian IAVs must adapt to overcome human cellular defenses. Although several protective barriers have been identified, such as receptor compatibility and polymerase adaptation, the full spectrum of human cellular defenses remains incompletely understood. Avian IAVs can effectively evade the human RNA-sensing pathway, yet they still exhibit limited replication capacity in human cells. This phenomenon suggests that other components of the human innate immune system may act as an additional barrier against avian IAVs.
RATIONALE
The DNA sensor adaptor stimulator of interferon genes (STING) is activated during IAV infection, and STING deficiency leads to increased IAV titers in vitro and in vivo. Thus, we investigated the role of STING in controlling viral replication in human cells and its potential role in restricting IAV cross-species transmission.
RESULTS
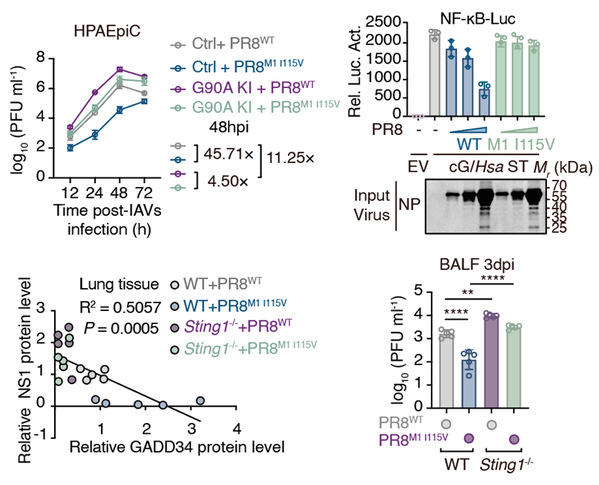
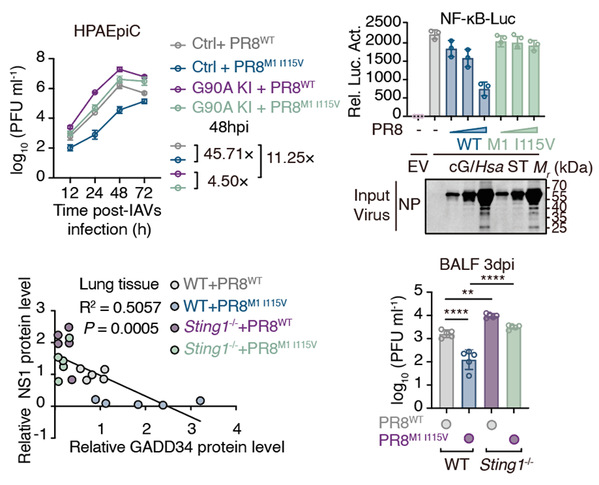
Using pharmacological and genetic approaches, we determined that STING restricted IAV by activating the nuclear factor κB (NF-κB) pathway. Further screening of a STING point-mutant library revealed that the Gly90 residue in STING was essential for activating NF-κB and restricting IAV replication. This finding was confirmed in primary human respiratory cells harboring endogenous Gly90 knock-in mutation and in point-mutant mice. We compared transcriptional changes induced by overexpressing wild-type STING with those initiated by the STING G90A (Gly90→Ala) mutant to identify candidate antiviral NF-κB–stimulated genes (NSGs) that we validated with a small interfering RNA screen. From this, we identified growth arrest and DNA damage–inducible protein 34 (GADD34), which restricted IAV replication in human cells. GADD34 was predominantly expressed in the human respiratory system and inhibited IAV polymerase activity independently of its canonical phosphatase function.
A screen of IAV-encoded proteins revealed that the matrix protein 1 (M1) protein encoded by human IAV mediated evasion of the human STING–NF-κB–GADD34 pathway. We found that the M1 protein derived from avian IAV was less effective than human IAV M1 in antagonizing human STING, although it retained full activity against avian STING. Sequence alignment identified a Val-to-Ile substitution at position 115 in the M1 protein that facilitated immune evasion specifically against human, but not avian STING, thereby enhancing the replicative fitness of avian-featured IAV in mammalian cells.
CONCLUSION
We propose that STING may act as a spillover barrier to prevent the avian-to-human transmission of IAVs. The evolution of residue 115 in the influenza virus M1 protein from Val to Ile may play a crucial role in circumventing this restriction and confers a replication advantage to the virus in human cells. Therefore, surveillance of the M1-115 variant can be used as a molecular marker for predicting IAV cross-species transmission risk. Additionally, developing agents that disrupt the M1-STING interaction, particularly against Ile115-bearing strains, could enhance preparedness for future influenza pandemics.

M1 protein evolution enables IAV to overcome the STING–NF-κB–GADD34 antiviral barrier.
The STING–NF-κB–GADD34 innate immune pathway acts as a host barrier against IAV in human cells. An isoleucine substitution at residue 115 of the human IAV M1 protein represents a key human-adaptive mutation that attenuates this antiviral defense, facilitating viral replication in human cells. Dashed lines of different thicknesses indicate relative levels of indirect regulation of the proteins shown. [Figure created with BioRender.com]
Abstract
Influenza pandemics are often traced back to the spillover of avian influenza A viruses (IAVs) to humans. However, barriers against IAV transmission remain elusive. We demonstrated human stimulator of interferon genes (STING) as a transmission barrier against IAVs. STING activated nuclear factor κB (NF-κB) and downstream NF-κB–stimulated genes (NSGs) through a specific domain. Among these NSGs, growth arrest and DNA damage–inducible protein 34 (GADD34) was crucial for IAV restriction. Some IAVs have evolved to evade activating human STING by mutating residue 115 in their matrix protein 1 (M1), which is essential for efficient viral replication in human respiratory cells. This barrier against the zoonotic threat of IAVs provides a tool for future investigations into the biological functions of the cyclic guanosine monophosphate–adenosine monosphosphate (cGMP-AMP) synthase (cGAS)–STING–NF-κB signaling pathway.