2026-03-18 マックス・プランク研究所
<関連情報>
- https://www.mpg.de/26263700/mystery-of-quinine-biosynthesis-solved
- https://www.nature.com/articles/s41586-026-10227-x
キナアルカロイドの生合成 Biosynthesis of cinchona alkaloids
Blaise Kimbadi Lombe,Tingan Zhou (周庭安),Gyumin Kang,Joshua C. Wood,John P. Hamilton,Klaus Gase,Yoko Nakamura,Ryan M. Alam,Ron P. Dirks,Lorenzo Caputi,C. Robin Buell &Sarah E. O’Connor
Nature Published:18 March 2026
DOI:https://doi.org/10.1038/s41586-026-10227-x
Abstract
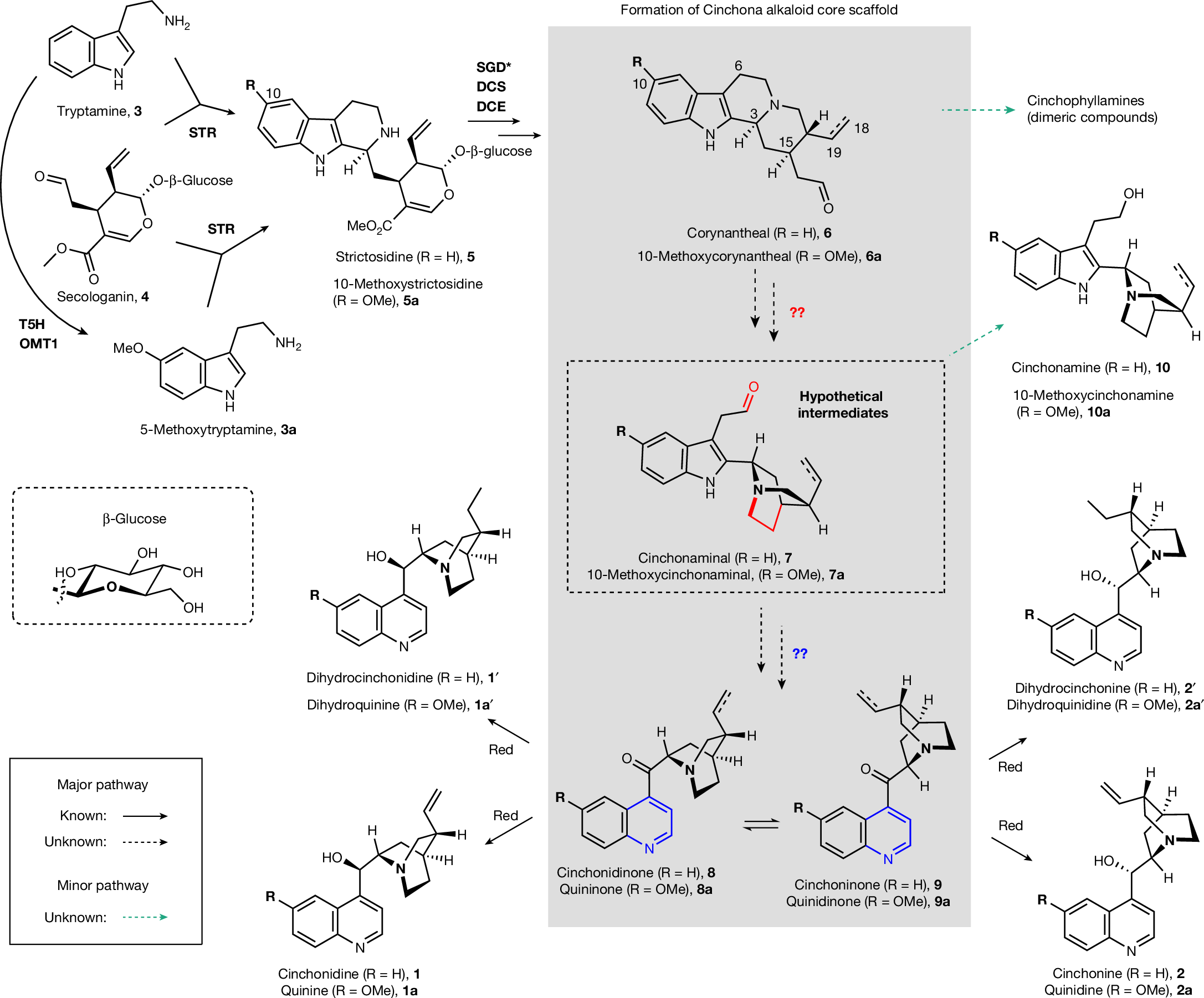
Cinchona alkaloids, which have been studied for more than 250 years, are plant-derived natural products that have collectively had a substantial impact in medicine and basic science1,2,3,4,5. Examples of cinchona alkaloids include quinine, a historically important antimalarial drug, and cinchonidine, a chiral catalyst widely used in process chemistry. However, it is still largely unknown how plants synthesize these well-known compounds. Here we report the discovery of genes responsible for the biosynthesis of the distinctive quinoline–quinuclidine scaffold of cinchona alkaloids. A combination of isotopic labelling, gene silencing, single-nucleus RNA sequencing and comparative transcriptomics revealed the involvement of several unexpected biosynthetic transformations. We also describe a previously unreported quaternary amine intermediate that is generated through an unusual enzymatic cyclization. We show that dihydroquini(di)none, dihydrocinchoni(di)none and cinchoni(di)none can be produced when these genes are heterologously expressed in Nicotiana benthamiana. Furthermore, we demonstrate that this N. benthamiana expression platform can convert non-native fluorinated and chlorinated tryptamine substrates into dihydrocinchoni(di)none analogues, which suggests that these biosynthetic enzymes can be leveraged to produce halogenated cinchona alkaloid derivatives. These discoveries uncover the long-standing mystery of how the cinchona alkaloid scaffold is biosynthesized and highlight prospects for access to these compounds through metabolic engineering approaches.