2026-04-14 テキサスA&M大学
<関連情報>
- https://stories.tamu.edu/news/2026/04/14/scientists-reverse-brain-aging-with-a-nasal-spray/
- https://isevjournals.onlinelibrary.wiley.com/doi/10.1002/jev2.70232
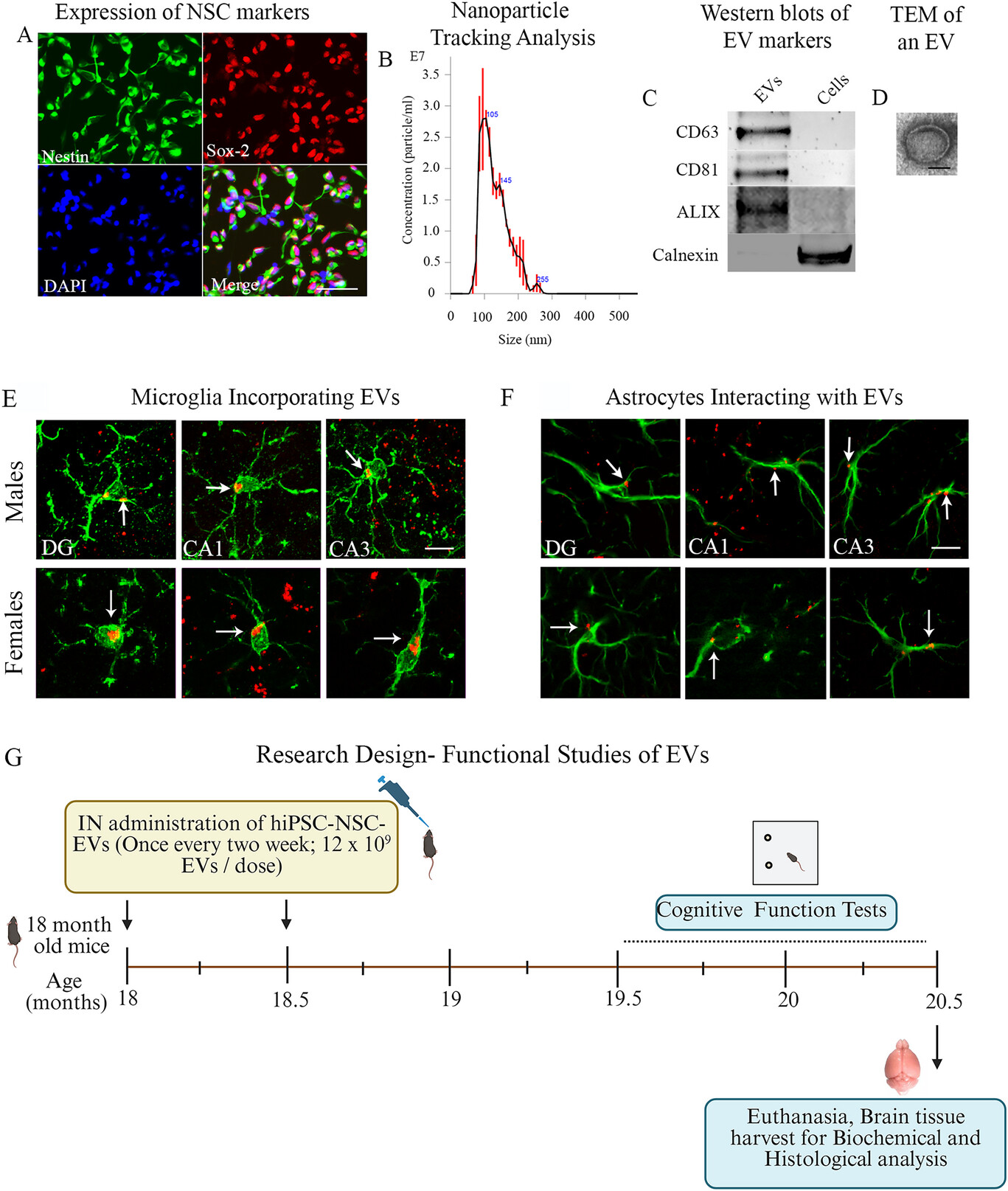
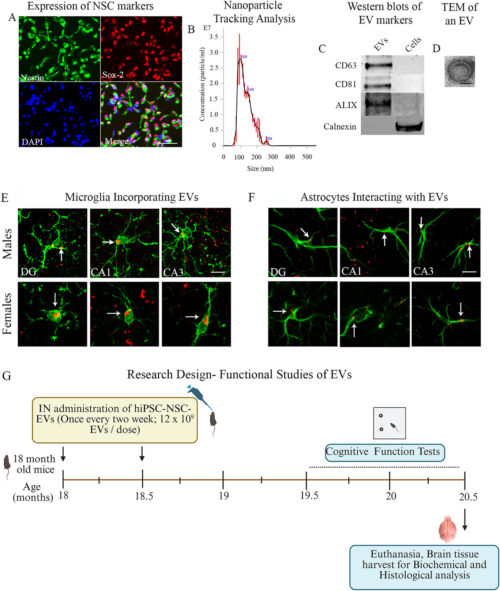
経鼻投与によるヒト神経幹細胞由来EV療法は、加齢した海馬における炎症性ミクログリア転写産物、NLRP3およびcGAS-STINGシグナル伝達を抑制できる Intranasal Human NSC-Derived EVs Therapy Can Restrain Inflammatory Microglial Transcriptome, and NLRP3 and cGAS-STING Signalling, in Aged Hippocampus
Leelavathi N. Madhu, Maheedhar Kodali, Shama Rao, Sahithi Attaluri, Raghavendra Upadhya, Goutham Shankar, Bing Shuai, Yogish Somayaji, Shruthi V. Ganesh, …
Journal of Extracellular Vesicles Published: 08 February 2026
DOI:https://doi.org/10.1002/jev2.70232
ABSTRACT
Neuroinflammaging, a moderate, chronic, and sterile inflammation in the hippocampus, contributes to age-related cognitive decline. Neuroinflammaging comprises the activation of the nucleotide-binding domain, leucine-rich repeat family, and pyrin domain-containing 3 (NLRP3) inflammasomes, and the cyclic GMP-AMP synthase (cGAS)-stimulator of interferon genes (STING) pathway that triggers type 1 interferon (IFN-1) signalling. Studies have shown that extracellular vesicles from human induced pluripotent stem cell-derived neural stem cells (hiPSC-NSC-EVs) contain therapeutic miRNAs that can alleviate neuroinflammation. Therefore, this study examined the effects of late middle-aged (18-month-old) male and female C57BL6/J mice receiving two intranasal doses of hiPSC-NSC-EVs on neuroinflammaging in the hippocampus at 20.5 months of age. Compared with animals receiving vehicle treatment, the hippocampus of animals receiving hiPSC-NSC-EVs exhibited reductions in astrocyte hypertrophy, microglial clusters, and oxidative stress, along with elevated expression of antioxidant proteins and genes that maintain mitochondrial respiratory chain integrity. Moreover, hiPSC-NSC-EVs therapy decreased the levels of various proteins involved in the activation of the NLRP3 inflammasome, p38/mitogen-activated protein kinase, cGAS-STING-IFN-1, and Janus kinase and signal transducer and activator of transcription signalling pathways. Furthermore, in vitro assays using genetically engineered RAW cells and hiPSC-NSC-EVs, with or without targeted depletion of specific miRNAs, demonstrated that miRNA-30e-3p and miRNA-181a-5p, both present in hiPSC-NSC-EVs, can significantly inhibit the activation of the NLRP3 inflammasome and the STING pathway, respectively. Additionally, single-cell RNA sequencing conducted 7 days post-treatment revealed that hiPSC-NSC-EVs induce widespread transcriptomic changes in microglia, including increased expression of numerous genes that enhance oxidative phosphorylation and reduced expression of abundant genes that drive multiple proinflammatory signalling pathways. These changes mediated by hiPSC-NSC-EVs were also associated with improved cognitive and memory function. Thus, intranasal hiPSC-NSC-EVs therapy in late middle age can effectively diminish proinflammatory microglial transcriptome and signalling cascades that drive neuroinflammaging in the hippocampus, contributing to better brain function in old age.