2026-04-28 国立がん研究センター,筑波大学,富山大学,日本臨床腫瘍研究グループ
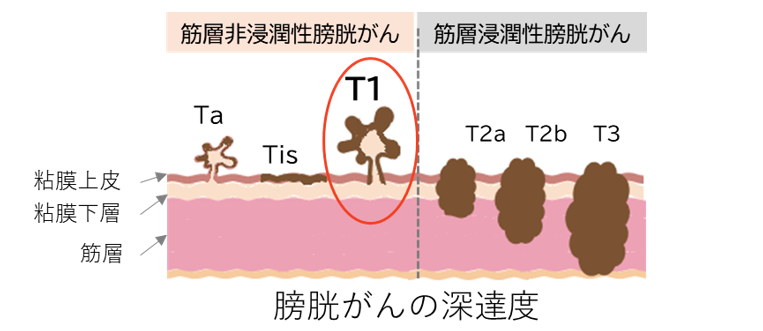
図1 膀胱がんの深達度
<関連情報>
- https://www.ncc.go.jp/jp/information/pr_release/2026/0428/index.html
- https://www.sciencedirect.com/science/article/pii/S030228382600014X
二次経尿道的切除術で陰性となった高悪性度T1膀胱癌に対する積極的監視療法と膀胱内BCG療法の比較:無作為化非劣性第3相JCOG1019試験 Active Surveillance Versus Intravesical Bacillus Calmette-Guérin for High-grade T1 Bladder Cancer with Negative Second Transurethral Resection: The Randomized Noninferiority Phase 3 JCOG1019 Trial
Hiroshi Kitamura, Taiji Tsukamoto, Yoshiyuki Kakehi, Junki Mizusawa, Taro Shibata, Keita Sasaki, Toshiki Tanikawa, Katsuyoshi Hashine, Kiyohide Fujimoto, Naoya Masumori, Takashi Kobayashi, Tomonori Habuchi, Takahiro Kimura, Mikio Sugimoto, Atsushi Takahashi, Hisanobu Adachi, Yoshiyuki Matsui, Shingo Hatakeyama, Akihiro Ito, Masatoshi Eto…Hiroyuki Nishiyama
European Urology Available online: 21 January 2026
DOI:https://doi.org/10.1016/j.eururo.2026.01.008
Abstract
Background and objective
We evaluated the noninferiority of active surveillance (AS) in comparison to intravesical bacillus Calmette-Guérin (BCG) in terms of recurrence and progression for patients with high-grade T1 (HG T1) bladder cancer at initial transurethral resection of the bladder (TURB) and no residual tumor at second TURB.
Methods
After initial evaluation, participants diagnosed with HG T1 bladder cancer who had undergone complete eradication of visible tumors underwent a second TURB. Those with specimens showing T0 were randomized to either AS or to intravesical BCG for 8 wk without maintenance therapy. The primary endpoint was invasive relapse–free survival (iRFS).
Key findings and limitations
In total, 513 participants were enrolled in the initial evaluation. After second TURB, 263 participants were enrolled and randomized. AS was noninferior to BCG in terms of iRFS (hazard ratio 0.69, 90% confidence interval 0.44–1.08; p = 0.001). Rates of adverse events were 50% and 90% for any grade, and in 3.1% and 3.8% for grade ≥3 events in the AS and BCG arms, respectively. The protocol treatment in the control arm was not the current standard.
Conclusions and clinical implications
In this highly selected patient population, AS was noninferior to eight-dose intravesical BCG induction therapy in terms of iRFS for T1 disease or deeper intravesical and/or extravesical recurrence. The safety profile of AS was better than that of BCG. These findings indicate that AS represents a potentially viable therapeutic strategy for selected patients with HG T1 bladder cancer for whom second TURB demonstrates the absence of residual disease.