2026-04-29 中国科学院深圳先進技術研究所(SIAT)
 Graphic summary.(Image by SIAT)
Graphic summary.(Image by SIAT)
<関連情報>
- https://english.siat.ac.cn/news/sr/202604/t20260414_1156177.html
- https://www.pnas.org/doi/10.1073/pnas.2523148123
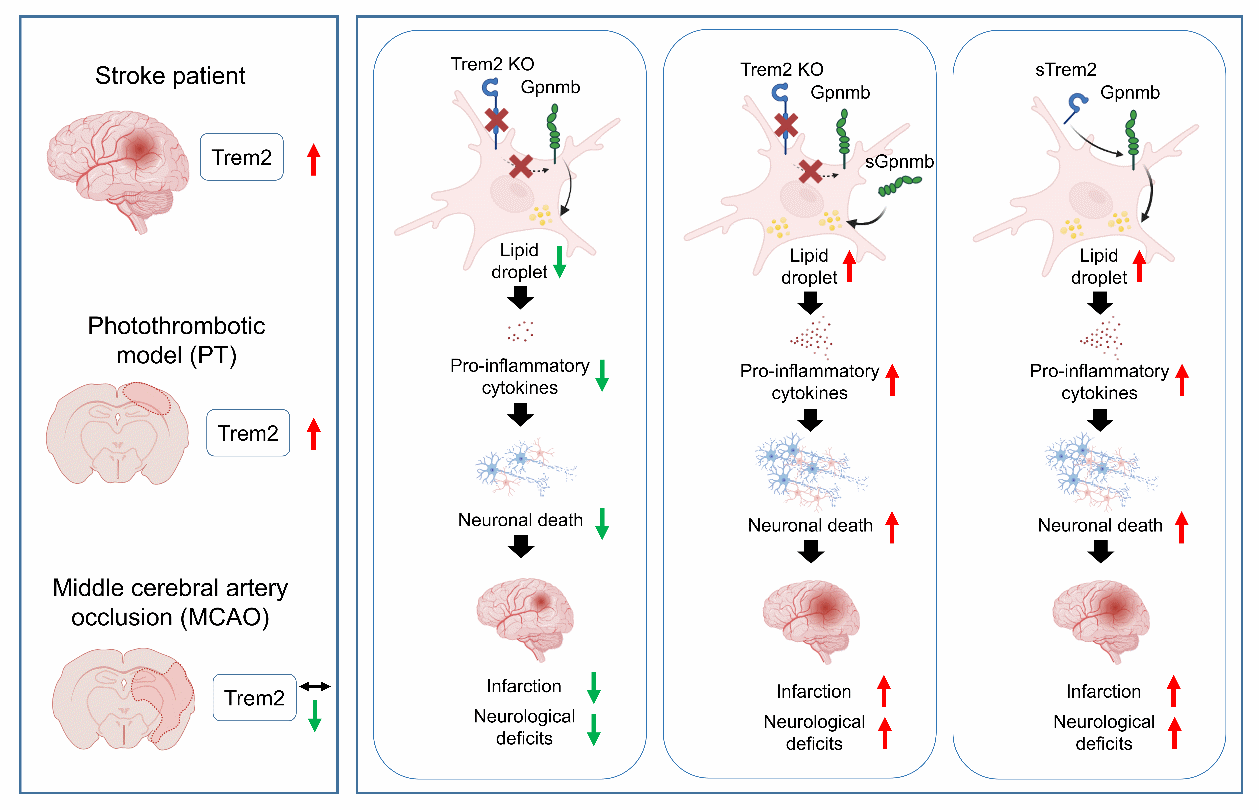
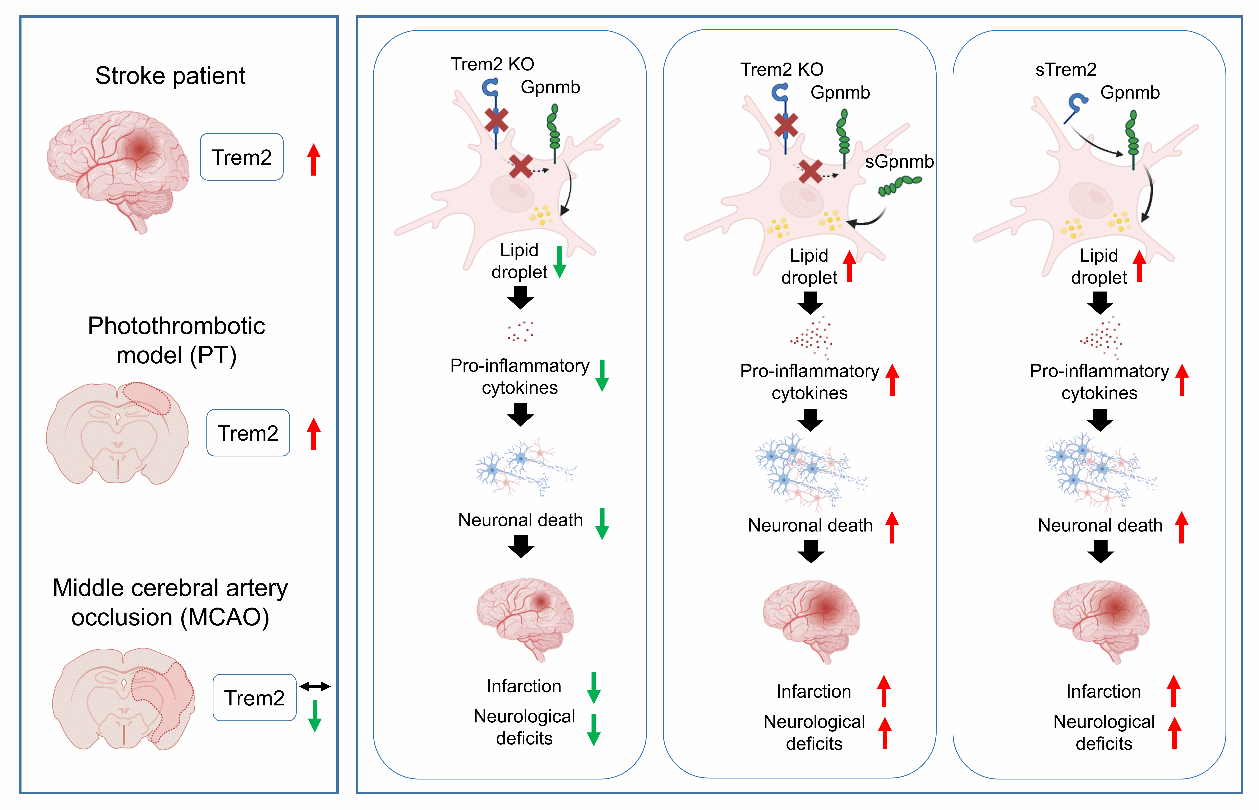
Trem2は光血栓性脳卒中モデルにおいてGpnmbを介して虚血性脳損傷を悪化させる Trem2 exacerbates ischemic brain injury through Gpnmb in a photothrombotic stroke model
Xuezhen Chen, Kunyu Li, Siyu Liu, +9 , and Xinzhou Zhu
Proceedings of the National Academy of Sciences Published:April 14, 2026
DOI:https://doi.org/10.1073/pnas.2523148123
Significance
Stroke is a leading cause of death and disability, with microglia-driven inflammation as a key contributor to brain injury. The microglial receptor Trem2 has shown inconsistent roles: protective in a classic stroke model but associated with worse outcomes when its soluble form (soluble Triggering receptor expressed on myeloid cells 2) is elevated in patients. Using a photothrombotic mouse model that better mirrors Trem2-mediated responses as observed in patients, we found that Trem2 drives sustained inflammation and exacerbates neuronal deficits. These results emphasize the importance of model selection in studying poststroke neuroinflammation and suggest Trem2 and its downstream pathways as potential therapeutic targets.
Abstract
Ischemic stroke is a major public health challenge, with microglia-mediated neuroinflammation exerting both protective and detrimental effects on neuronal survival. The Triggering receptor expressed on myeloid cells 2 (Trem2), predominantly expressed by microglia, has been reported to confer neuroprotection in the middle cerebral artery occlusion (MCAO) model. Paradoxically, in patients, elevated plasma soluble Trem2 (sTrem2) levels correlate with increased risk and poor outcomes. To test the impact of Trem2 function in the context of stroke, we utilized the photothrombotic stroke model which elicited strong Trem2 upregulation, a clinical feature which is not mimicked in MCAO models. Trem2 depletion reduced infarction volume, suppressed proinflammatory cytokine production, preserved neuronal survival, and lessened motor and neurological impairment. Conversely, intracerebral administration of sTrem2 exacerbated neuronal loss, amplified inflammation, and worsened neurological deficits. Integrated mouse-human transcriptomic analyses identified glycoprotein nonmetastatic B (Gpnmb) as a conserved downstream effector of Trem2. Soluble Gpnmb (sGpnmb) administration abolished the protective effects of Trem2 depletion, promoting microglial activation, lipid accumulation, and neuronal damage. Additionally, plasma sTrem2 and sGpnmb levels were elevated in stroke patients, positively correlated, and may serve as biomarkers of poor prognosis. These findings uncover a detrimental role for Trem2 in ischemic stroke, provide mechanistic insight into the link between sTrem2 and poor clinical outcomes, and identify the Trem2–Gpnmb axis as a potential therapeutic target to mitigate poststroke neuroinflammation.