2026-03-11 テキサス大学オースティン校
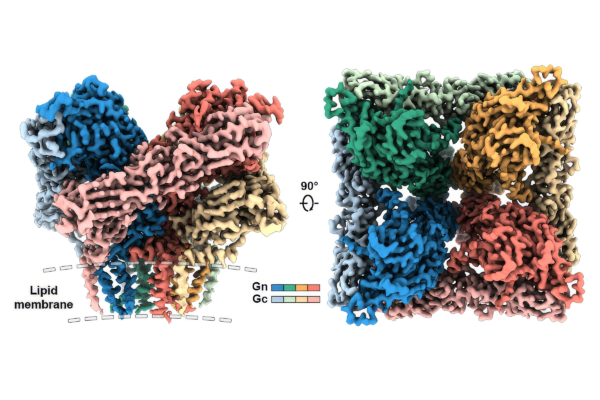
This is the highest resolution molecular structure ever constructed of a key protein complex on the surface of the Andes virus, called the Gn-Gc tetramer, that enables it to infect cells.
<関連情報>
- https://news.utexas.edu/2026/03/11/scientists-map-deadly-hantavirus-bringing-treatments-one-step-closer/
- https://www.cell.com/cell/fulltext/S0092-8674(26)00117-0
ハンタウイルス糖タンパク質テトラマーの高解像度in situ構造 High-resolution in situ structures of hantavirus glycoprotein tetramers
Luqiang Guo (郭鲁强) ∙ Elizabeth McFadden ∙ Megan M. Slough ∙ … ∙ Jesse H. Erasmus ∙ Kartik Chandran ∙ Jason S. McLellan
Cell Published:February 27, 2026
DOI:https://doi.org/10.1016/j.cell.2026.01.030
Highlights
- Polarized distribution of ANDV Gn–Gc glycoprotein tetramers on rVSV surface
- Complete single-particle cryo-EM workflow for lattice-forming viral glycoproteins
- Structures of ANDV Gn–Gc tetramer, dimers of tetramers, and Fab complex on eVLPs
- RepRNA encoding ANDV-VLPs elicits neutralizing antibody titers in mice
Summary
New World hantaviruses cause severe infections in humans. Previous structural studies have advanced our understanding of hantavirus glycoprotein architecture; however, the lack of high-resolution structures of the glycoprotein tetramer and its lattice organization has limited mechanistic insights into viral assembly and entry. Here, we leveraged a virus-like particle (VLP) system to establish a cryo-electron microscopy workflow for lattice-forming viral glycoproteins. This enabled the determination of a 2.35 Å resolution structure of the membrane-embedded Andes virus (ANDV) glycoprotein tetramer as well as the structures of dimers of tetramers and a complex with antibody ADI-65534. These structures reveal previously uncharacterized features of glycoprotein organization, stability, and pH sensing. The immunization of mice with self-amplifying replicon RNA (repRNA) encoding ANDV-VLPs elicited high levels of glycoprotein-binding antibodies but equivalent titers of neutralizing antibodies compared with the repRNA-encoded native ANDV glycoprotein complex. These findings advance our understanding of hantavirus glycoprotein assemblies, laying a foundation for structure-based vaccine design.