2026-04-30 京都大学
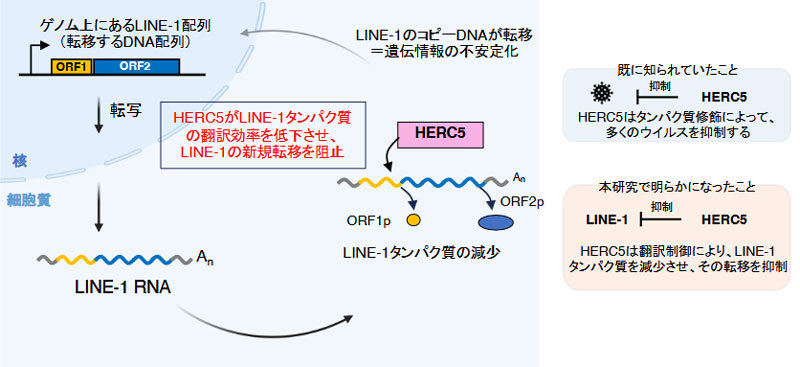
HERC5によるLINE-1の抑制メカニズム:ゲノム上を移動(転移)するLINE-1は、自身の転移に必要なタンパク質(ORF1p)を作り出す。
(図作成:西森奎、Created with BioRender.com)
<関連情報>
- https://www.kyoto-u.ac.jp/ja/research-news/2026-04-30-1
- https://academic.oup.com/nar/article/54/8/gkag334/8664513
インターフェロン誘導型遺伝子産物HERC5はISG化非依存的な機構により、ヒトLINE-1レトロトランスポゾンの転移を抑制する The interferon-stimulated gene product HERC5 inhibits human LINE-1 retrotransposition with an ISGylation-independent mechanism
Kei Nishimori,Ahmad Luqman-Fatah,Yuzo Watanabe,Mari Takahashi,Takuhiro Ito,Fuyuki Ishikawa,Tomoichiro Miyoshi
Nucleic Acids Research Published:30 April 2026
DOI:https://doi.org/10.1093/nar/gkag334
Abstract
Mobilization of Long INterspersed Element-1 (LINE-1 or L1) compromises genome stability and can cause sporadic genetic diseases. Accordingly, cells have evolved multiple mechanisms to restrict L1 retrotransposition. Several interferon-stimulated genes (ISGs) that interact with cytoplasmic L1 ribonucleoproteins (RNPs), which contain the L1-encoded proteins ORF1p and ORF2p, have been identified as suppressors of L1 retrotransposition. We previously reported that the ISG protein HECT and RLD domain containing E3 ubiquitin-protein ligase 5 (HERC5) efficiently inhibits L1 retrotransposition. While HERC5 is known to restrict numerous viruses through ISGylation, how HERC5 inhibits L1 remains to be elucidated. Here, we show that HERC5 inhibits L1 retrotransposition through an ISGylation-independent mechanism. HERC5 interacts with L1 RNA and selectively reduces ORF1p levels in a manner that requires the full-length ORF1p expression. We further demonstrate that HERC5 decreases L1 translation efficiency and alters L1 RNP composition. Our comparative analysis further suggests that HERC5 may have acquired its L1-inhibitory function during the evolution of the small HERC family. These findings uncover a previously unidentified mechanism by which an ISG protein associates with and inhibits L1 and suggest a role for HERC5 as an evolutionarily adapted restriction factor that expands the repertoire of cellular defenses against retrotransposons.