2026-03-25 パデュー大学
<関連情報>
- https://ag.purdue.edu/news/2026/03/protein-modification-discovery-opens-cancer-therapy-possibilities.html
- https://www.nature.com/articles/s41557-026-02093-x
D-およびL -2-ヒドロキシグルタル酸によるキラル依存性タンパク質修飾の発見 Discovery of chirally dependent protein modifications by D- and L-2-hydroxyglutarates
Zheng Zhang,Yi-Kai Liu,Zhuojun Luo,Meng-Ju Wu,Claudia N. Evans,Zihan Qu,Fanglei Xue,Zijian Wang,Lia Stanciu,Zhong-Yin Zhang,Elizabeth I. Parkinson,Nabeel Bardeesy & W. Andy Tao
Nature Chemistry Published:17 March 2026
DOI:https://doi.org/10.1038/s41557-026-02093-x
Abstract
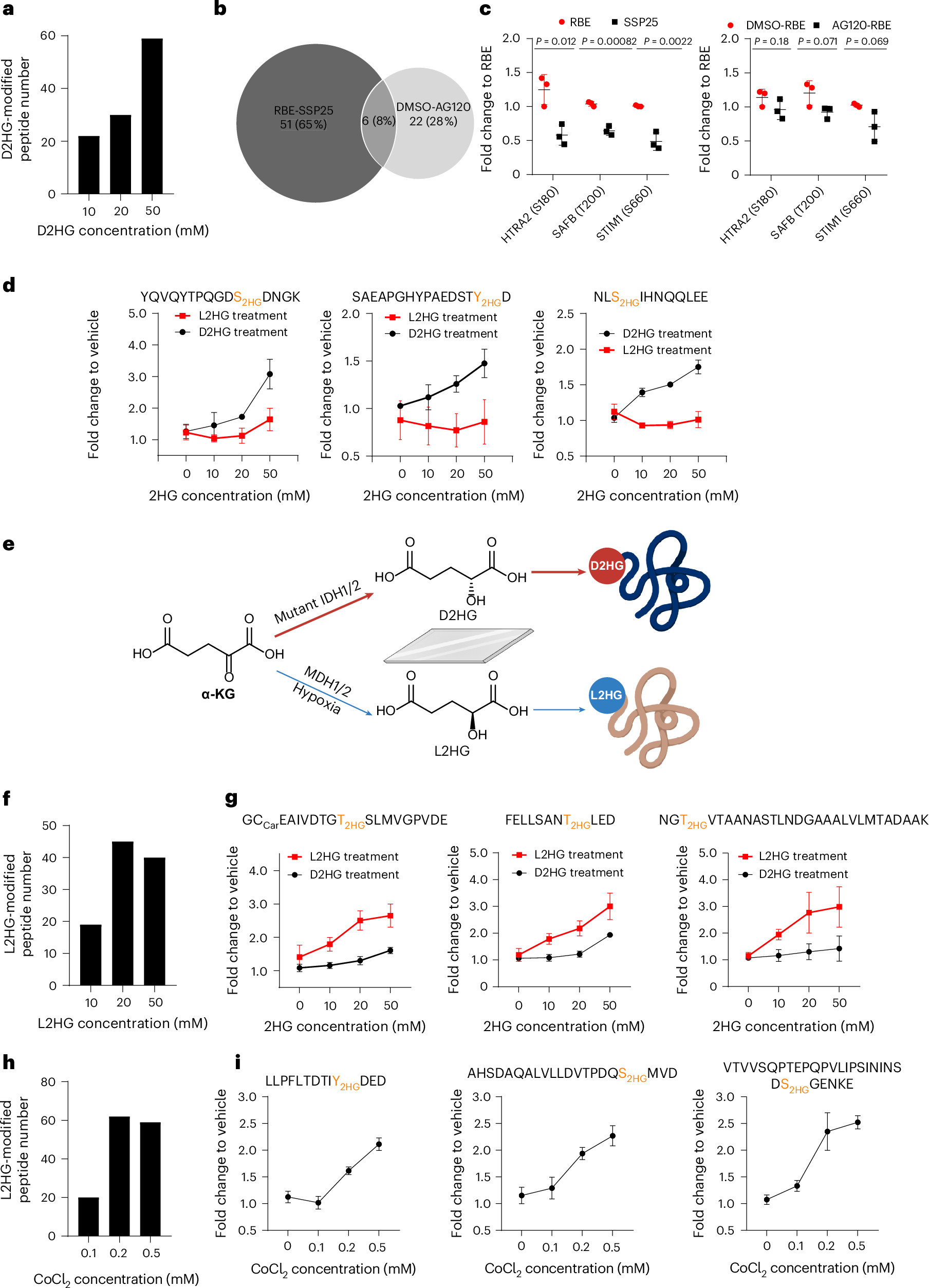
Mutations in isocitrate dehydrogenase 1 (IDH1) and 2 (IDH2) are common in multiple types of human cancer and cause accumulation of the oncometabolite D-2-hydroxyglutarate (D2HG) instead of α-ketoglutarate, driving cancers like gliomas and acute myeloid leukaemia by blocking cell differentiation and promoting tumour growth. Here we discovered protein O-2-hydroxyglutarylation by D2HG using chemical proteomics and further revealed distinct chiral preferences for D2HG and L-2-hydroxyglutarate (L2HG) modifications. D2HG modifications are upregulated in IDH-mutant cells or upon D2HG treatment, while L2HG modifications increase under hypoxic conditions or following L2HG treatment. Notably, two kinases MRCKA and SLK are modified by D2HG and L2HG, respectively, and confirmed by synthetic peptide standards. Phosphoproteomics revealed reduced phosphorylation of MRCKA and SLK substrates, suggesting crosstalk between D/L-2HG modification and kinase activity. These findings highlight distinctive roles of D/L-2HG modifications in cancer progression and suggest potential avenues for therapeutic targeting of oncometabolite-induced post-translational modifications.