2026-04-21 東北大学
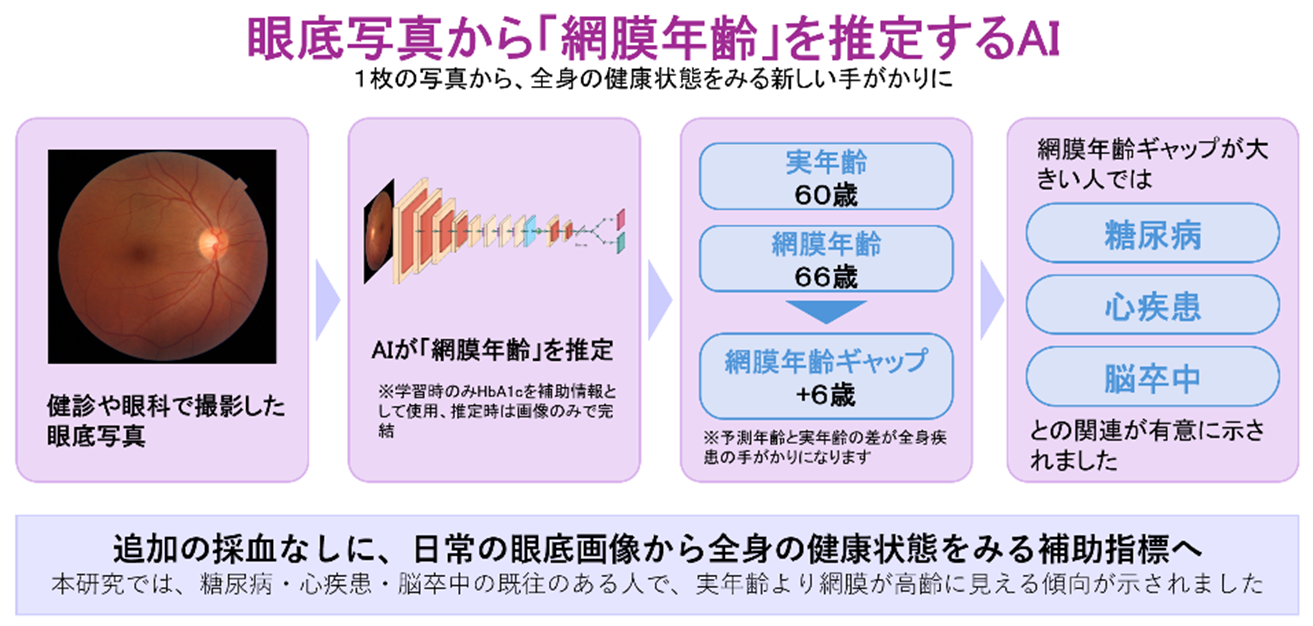
図1.眼底写真から「網膜年齢」を推定するAI
<関連情報>
- https://www.tohoku.ac.jp/japanese/2026/04/press20260421-01-fundus.html
- https://www.tohoku.ac.jp/japanese/newimg/pressimg/tohokuuniv-press20260421_01_fundus.pdf
- https://www.nature.com/articles/s43856-026-01573-y
眼底に基づくマルチタスク学習による高精度網膜年齢予測により、全身疾患の影響が明らかになる High-accuracy retinal age prediction via fundus-based multitask learning reveals the effect of systemic disease
Takahiro Ninomiya,Akiko Hanyuda,Naoki Kiyota,Parmanand Sharma,Yukun Zhou,Siegfried K. Wagner,Keita Suzuki,Takanari Nozaki,Takehiro Miya,Naoki Takahashi,Kazuko Omodaka,Noriko Himori,Yoichi Ichikawa,Pearse A. Keane & Toru Nakazawa
Communications Medicine Published:08 April 2026
DOI:https://doi.org/10.1038/s43856-026-01573-y Unedited version
Abstract
Background
Accurate estimation of the retinal age, defined as the age predicted from fundus photographs by a deep-learning model trained on chronological age, provides a non-invasive biomarker of biological ageing and disease risk.
Methods
In this study, we trained an ensemble multitask learning model that integrates fundus photographs with glycated haemoglobin using 50,595 quality-controlled fundus photographs from 27,214 disease-free adults and validated it on an independent set of 7288 additional images from disease-free adults. Model performance was evaluated using mean absolute error. Prediction uncertainty was quantified by calculating the standard deviation across ensemble predictions for each eye, and eyes were stratified based on this standard deviation.
Results
Here we show that the model achieves mean absolute errors of 2.78 years in internal validation and 3.39 years and 8.63 years in two external cohorts comprising 135 and 4992 eyes, respectively. Eyes with ensemble standard deviations below the median demonstrate improved age-prediction accuracy (mean absolute error: 2.46 years internally; 2.87 years in the primary external cohort). In a systemic disease cohort of 8467 individuals, the retinal age gap (predicted minus chronological age) is significantly higher in participants with diabetes, cardiac disease, or stroke after adjustment for age and sex, indicating older-appearing retinas and supporting the biological relevance of retinal age.
Conclusions
Retinal age derived from a single fundus photograph provides a scalable and clinically deployed biomarker of biological ageing. This approach may enable opportunistic screening for cardiometabolic and other ageing-related diseases in other routine ocular imaging workflows.
Plain language summary
Retinal photographs are images of the inner, back surface of the eye and are routinely taken in eye clinics. Ageing alters the retinal appearance. We built a computational model that estimates the age of the retina from a single retinal photo. The system was trained on 50,595 images from 27,214 disease‑free adults. Predictions of age were most accurate when high‑quality images were used and remained accurate when tested on additional images. A large gap between a person’s actual age and the estimated retinal age was found more in people with diabetes, heart disease or stroke. This tool could therefore be helpful to assess cardiometabolic health during routine eye appointments.


