2026-04-23 京都大学iPS細胞研究所
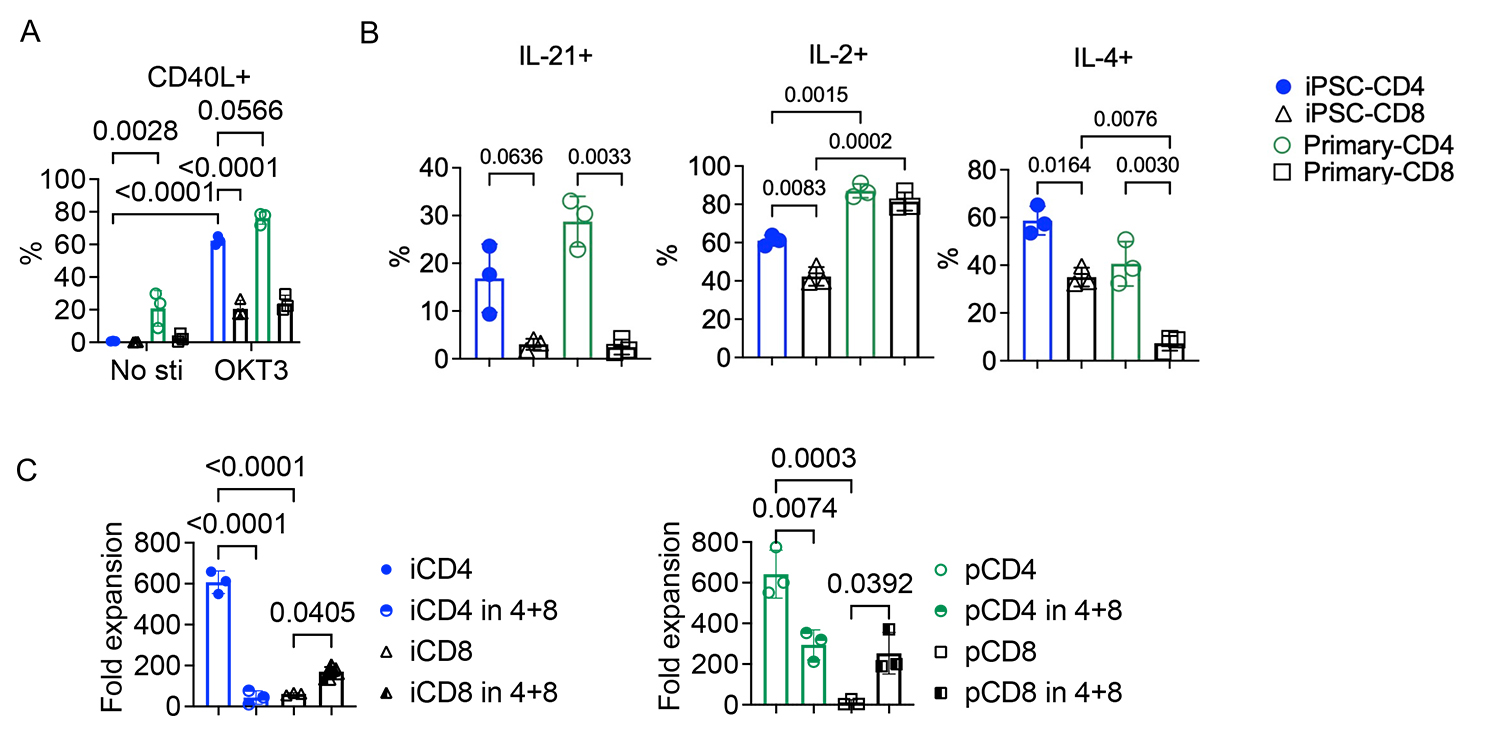
図1. iCD4+T細胞のヘルパー表現型および機能
<関連情報>
- https://www.cira.kyoto-u.ac.jp/j/pressrelease/news/260423-100000.html
- https://link.springer.com/article/10.1186/s41232-025-00402-4
適応型CAR-iPSC-CD4⁺ T細胞は、持続的なALL制御においてCD8⁺ T細胞よりも優れた性能を示す Adaptive-like CAR-iPSC-CD4⁺ T cells outperform CD8⁺ counterparts in sustained ALL control
Qingyi Guo,Chaoqi Zhang,Bo Wang,Shoichi Iriguchi,Akihiro Ishikawa,Atsutaka Minagawa,Tomoko Ishii,Yohei Kawai & Shin Kaneko
Inflammation and Regeneration Published:03 January 2026
DOI:https://doi.org/10.1186/s41232-025-00402-4
Abstract
Background
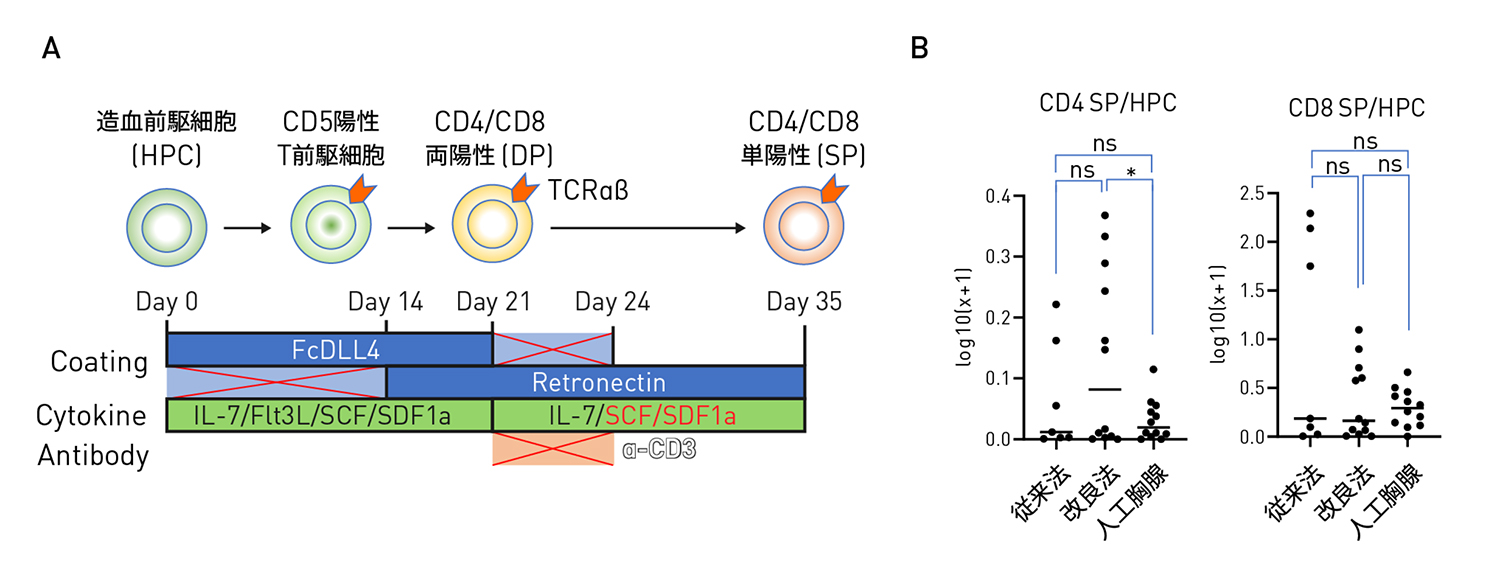
Induced pluripotent stem cell (iPSC)–derived T cells offer a renewable source for off-the-shelf immunotherapy. With the advent of the artificial thymic organoid (ATO) method, the in vitro differentiation of CD4+ T cells from iPSCs has also become feasible. CD4⁺ T cells have shown superior longevity, resistance to exhaustion, and helper functions in primary settings, but whether iPSC-derived CD4⁺ T cells retain these features remains unclear.
Methods
In this study, CD4⁺ T cells were differentiated from human iPSCs using the ATO system. Primary T cells served as controls to evaluate the phenotypic and activation features of iPSC-derived CD4⁺ and CD8⁺ T cells. To assess antitumor function, we generated CD19-BBζ CAR-iPSC-T cells and employed a hematologic malignancy model using NALM6 acute lymphoblastic leukemia (ALL) cells. Both short-term and long-term cytotoxicity assays were conducted to compare iPSC-derived CD4⁺ and CD8⁺ T cells in terms of killing efficiency, cytokine secretion, persistence, exhaustion phenotype, and proliferative capacity. The helper function of iPSC-derived CD4⁺ T cells toward CD8⁺ T cells was further evaluated by Ki-67 staining and proliferation assays. Statistical analyses were performed using GraphPad Prism.
Results
Our study demonstrated that iPSC-derived CD4⁺ T cells exhibited both helper- and cytotoxic-like features. Compared with iPSC-derived CD8⁺ T cells or CD4⁺/CD8⁺ mixtures, iPSC-derived CD4⁺ T cells showed superior proliferation, cytokine secretion, and sustained cytotoxicity following CAR transduction. They also promoted the expansion of iPSC-derived CD8⁺ T cells and displayed helper-like functions with increased resistance to exhaustion.
Conclusions
Although not identical to primary CD4⁺ T cells, iPSC-derived CD4⁺ T cells recapitulated key functional advantages, especially sustained antitumor activity, supporting their value as a renewable, off-the-shelf source for next-generation CAR-T therapies.