2026-05-14 東京農工大学
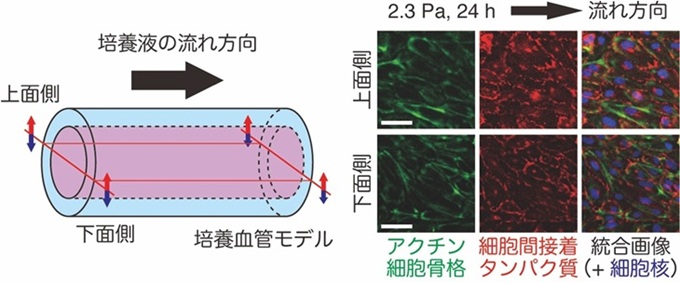
図1:培養血管モデル内部を単層で覆う血管内皮細胞の免疫蛍光染色画像。
図は(Okuno et al, 2026. BMC Methods, 3, 16)を改変して吉野らにより作成。
<関連情報>
- https://www.tuat.ac.jp/outline/disclosure/pressrelease/2026/20260514_01.html
- https://link.springer.com/article/10.1186/s44330-026-00072-9
細胞培養PDMS血管モデルにより、インプラントデバイスの配置が可能になる Cell-cultured PDMS vascular model to allow placement of implant devices
Taku Okuno,Kazuyo Ito,Kenichi Funamoto & Daisuke Yoshino
BMC Methods Published:12 May 2026
DOI:https://doi.org/10.1186/s44330-026-00072-9
Abstract
Background
A stent maintains normal blood flow by expanding a stenotic artery from the inside. However, current stents are mechanically sub-optimized and can exert excessive forces on the vascular wall, leading to inflammation, late thrombosis, and in-stent restenosis. Optimizing the mechanical performance of stents requires not only reproducing the mechanical field within the stented vessel but also evaluating endothelialization, which serves as a key biological indicator of vascular neointimal formation. To this end, this study aimed to develop a three-dimensional in vitro stent endothelialization model that enables quantitative evaluation of endothelial responses under physiologically relevant mechanical conditions and to provide detailed fabrication protocols for its construction.
Methods
Polydimethylsiloxane (PDMS) was used to mimic the adventitial structure of arteries. Human carotid artery endothelial cells (HCtAECs) were then seeded on the luminal surface and cultured for 24 h to form a confluent monolayer (intima). The constructed model was installed in the flow-exposure culturing system, and hemodynamic stimuli (two types of shear stress (SS); 0.5 Pa and 2.3 Pa) were applied to the HCtAECs inside to reproduce the physiological state of blood vessels. A self-expanding stent was then placed in the model during perfusion culture to evaluate in-stent endothelialization under controlled flow conditions.
Results
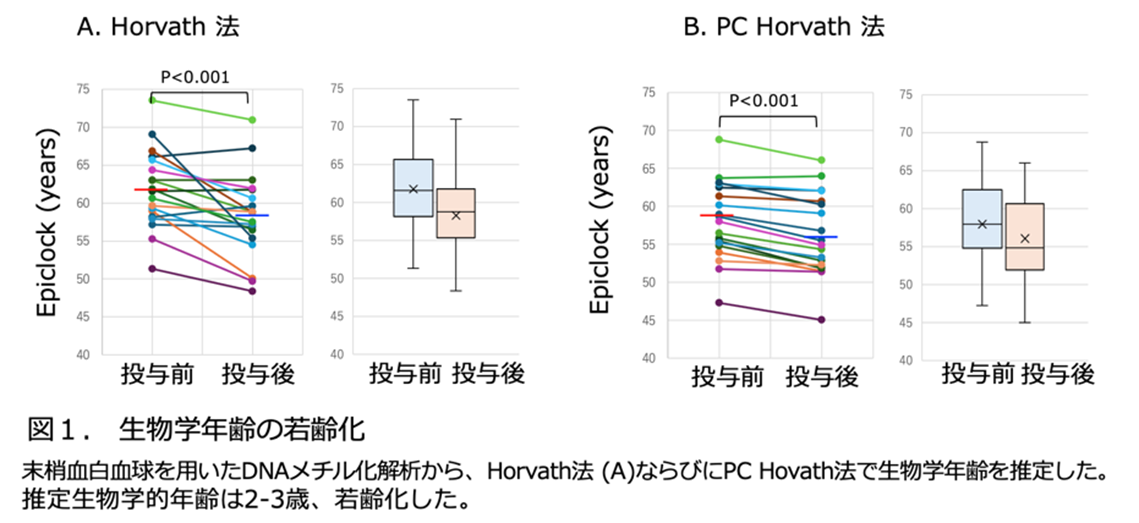
We examined the performance of the developed model based on quantitative evaluations of endothelial morphology in response to SS and in-stent endothelialization. Exposure to SS for 24 and 48 h caused endothelial orientation and elongation in the direction of flow, confirming the physiological responses of blood vessels. Furthermore, spatial and temporal analyses of in-stent endothelialization confirmed that the model can reproduce key biological processes associated with vascular neointimal formation in the presence of mechanical stimulation.
Discussion
The present model successfully integrates the mechanical and biological aspects of stent–vessel interaction, providing a reproducible platform for evaluating in-stent endothelialization under physiologically relevant conditions. This system can serve as a powerful tool not only for the quantitative assessment of endothelial dynamics but also for guiding the optimization of mechanical forces in stented blood vessels. Consequently, it offers a foundation for designing next-generation stents that promote rapid endothelialization and reduce the risk of restenosis and thrombosis.