2026-03-25 京都大学

研究概要図(イラスト:Kanon Tanaka)
<関連情報>
- https://www.kyoto-u.ac.jp/ja/research-news/2026-03-25-1
- https://www.kyoto-u.ac.jp/sites/default/files/2026-03/web_2603_Murakami-c78c6971a23d5548b0f443ffb2dc8f72.pdf
- https://pubs.acs.org/doi/full/10.1021/acsmeasuresciau.5c00193
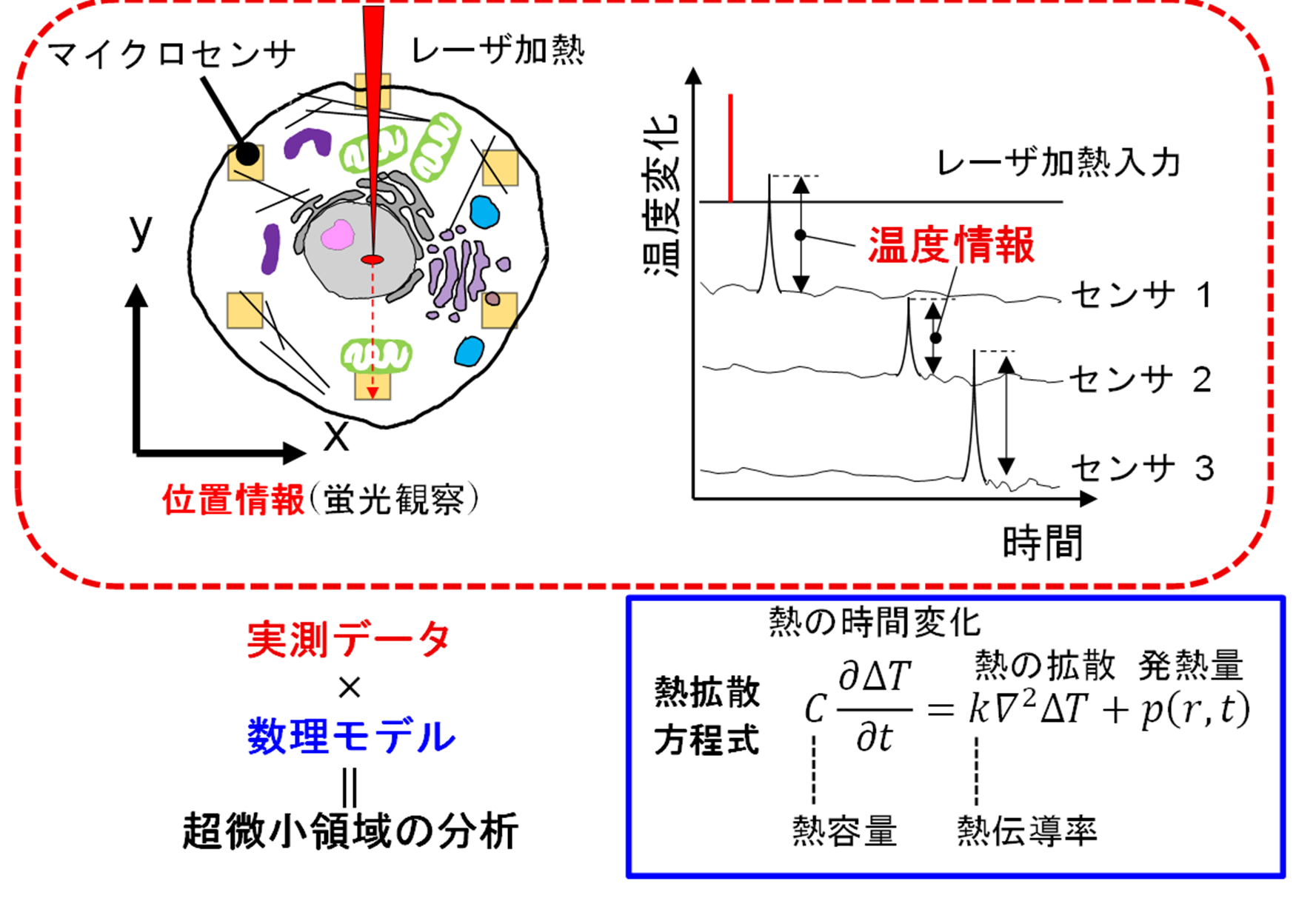
¹H検出型77Se/125Te NMRを用いたグルタチオントリカルコゲナイドにおけるカルコゲン間結合の解析 Exploring Chalcogen Connection in Glutathione Trichalcogenides via 1H-Detected 77Se/125Te NMR
Kazuma Murakami,Keisuke Tao-Kakuyama,Thi Hong Van Nguyen,Katsutoshi Nishino,and Takaaki Akaike
ACS Measurement Science Au Published: March 15, 2026
DOI:https://doi.org/10.1021/acsmeasuresciau.5c00193
Abstract
Trichalcogenides with linearly catenated chalcogen atoms (S, Se, and Te), exemplified by glutathione trisulfide, are increasingly recognized as key regulators of biological redox processes. However, their robust structural characterization remains limited because the NMR-active sulfur isotope (33S, I = 3/2) is low-abundance and quadrupolar. The examination of chalcogen connection in heterologous trichalcogenides remains highly challenging. Recent studies have examined 77Se and 125Te as replacements for sulfur and applied 77Se/125Te NMR in biomolecules. In this study, we report the in situ synthesis of heterologous trichalcogenides of glutathione disulfide (GS-SG) by inserting a single chalcogen atom into a disulfide bond using chalcogen donors in a conventional NMR tube. The NMR-active isotopes 77Se and 125Te (I = 1/2) provide broad chemical-shift ranges and high sensitivity to structural and dynamic changes, enabling multinuclear NMR. 1H DOSY and quantitative 1H NMR confirm a single major species (>95% by NMR) and 1H-detected 77Se/125Te heteronuclear multiple-bond correlation experiments demonstrate long-range coherence transfers. Heavy chalcogen insertion causes greater structural perturbation, making Se more suitable than Te. Density functional theory calculation revealed a smaller highest occupied molecular orbital–lowest unoccupied molecular orbital gap for GS-Te-SG, indicating its lower thermodynamic stability than GS-Se-SG. Consistent results were observed in the cystine-based experiments. To the best of our knowledge, for the first time, this study demonstrated that Se or Te can be inserted into the central site of disulfide bonding. In the biological assays, glutathione derivatives incorporating heavier chalcogens exhibit antiradical activities. This in situ synthetic and analytical framework expands the chemical space of biologically relevant chalcogenides.