2026-03-27 浙江大学(ZJU)
<関連情報>
- https://www.zju.edu.cn/english/_t874/2026/0327/c19573a3145038/page.htm
- https://www.vita-journal.com/vita/EN/10.15302/vita.2026.03.0017
胎児肝臓におけるTRPV4のアセチル化は、低血糖によるmTORC1阻害を防ぎ、胎児の発育を保護する TRPV4 acetylation in prenatal liver prevents low glucose-induced inhibition of mTORC1 and safeguards fetal development
Chen-Jie Zhang, Chuan-Jin Yu, Yi Cheng, Jie-Xue Pan, Long-Yun Ye, Yun-Hui Tang, Xue-Yun Qin, Zhong-Liang Lin, Ya-Ying Wu, Ci-Xiong Zhang, Jian-Feng Wu, Yi-Ran Zhao, Ke-Xin Zou, Chen-Song Zhang, Guo-Lian Ding, Sheng-Cai Lin, He-Feng Huang
Vita Published: March 13, 2026
DOI:https://doi.org/10.15302/vita.2026.03.0017
ABSTRACT
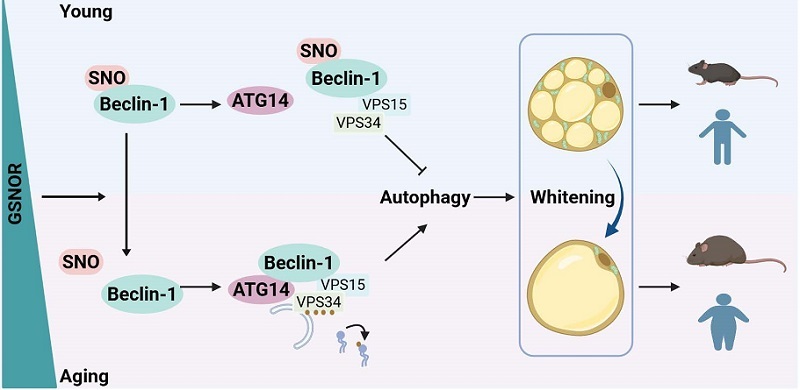
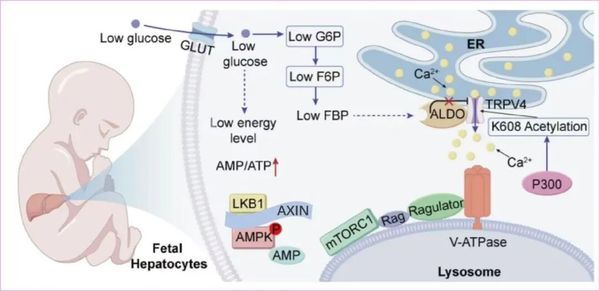
During the prenatal development of mammals, the fetus resides in a low-glucose environment compared to the mother, a physiological gradient essential for transplacental glucose transfer. In spite of low glucose, it is essential to maintain an active anabolic metabolism to support rapid fetal growth. However, how fetal tissues sustain this anabolic metabolism remains unknown. Here, we observed surprisingly that in the fetal liver, mTORC1 — a central metabolic regulator that promotes anabolism — remains active in low glucose, a condition where it is typically inhibited, although AMPK, another metabolic regulator that promotes catabolism, is effectively activated in an AMP-dependent manner. Mechanistically, we discovered that the upstream regulator of mTORC1, TRPV4 (a key TRPV channel in the liver), is acetylated at K608, disabling its inactivation by fructose-1,6-bisphosphate (FBP)-unoccupied aldolase and blunting the inhibition of mTORC1 that usually occurs in low glucose. Expression of a non-acetylatable TRPV4-K608R mutant in fetal hepatocytes restores glucose sensitivity and inhibits mTORC1, and liver-specific expression of this mutant during embryonic development impairs hepatic anabolism, reduces fetal protein synthesis, and leads to intrauterine growth restriction or even death. Our findings reveal that resistance of mTORC1 to low glucose-induced inhibition mediated by TRPV acetylation acts as a safeguard for normal fetal development.