2026-04-29 ミュンヘン大学(LMU)
<関連情報>
- https://www.lmu.de/en/newsroom/news-overview/news/one-molecule-two-effects-a-new-drug-concept-to-treat-obesity-and-type-2-diabetes-70cf6241.html
- https://www.nature.com/articles/s41586-026-10427-5
GLP-1R–GIPR–PPARα/γ/δ五重アゴニストはマウスの肥満と糖尿病を改善する GLP-1R–GIPR–PPARα/γ/δ quintuple agonism corrects obesity and diabetes in mice
Daniela Liskiewicz,Aaron Novikoff,Ahmed Khalil,Seun Akindehin,Jonathan E. Campbell,Pietra Candela,Russell L. Castelino,Callum Coupland,Maxime Culot,W. Scott Dodson,Jonathan D. Douros,Hannes Embring,Annette Feuchtinger,Brian Finan,Cristina Garcia-Caceres,Xiao-Bing Gao,Fabien Gosselet,Gerald Grandl,Robert M. Gutgesell,Daniel T. Haas,Martin Jastroch,Ezgi Karaoglu,Pamela Kakimoto,Anna Cristina Kaltenbach,… Timo D. Müller
Nature Published:29 April 2026
DOI:https://doi.org/10.1038/s41586-026-10427-5
Abstract
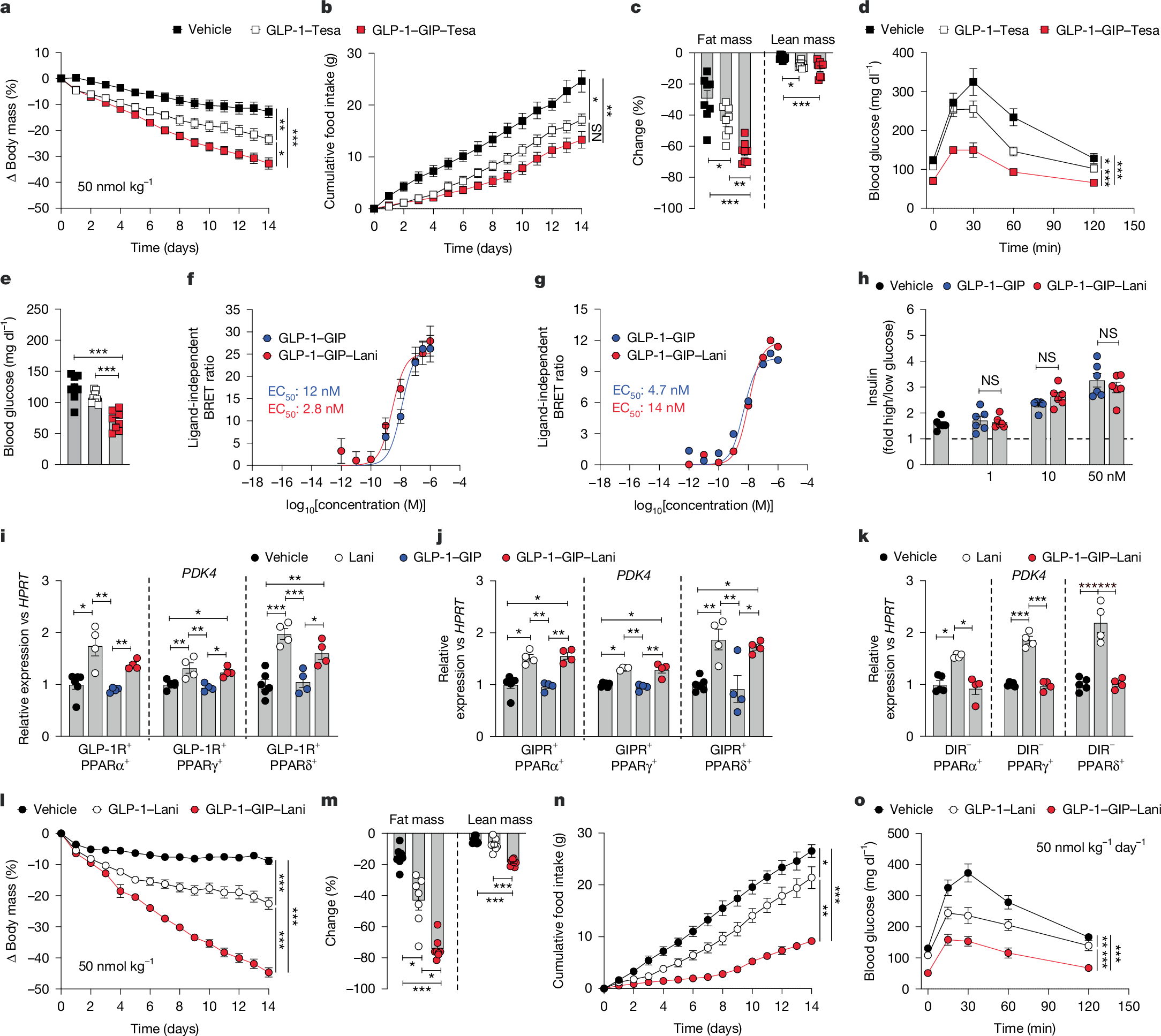
There are increasing numbers of effective drugs to improve obesity-linked metabolic dysfunction; GLP-1R–GIPR co-agonism is effective in the management of obesity and type 2 diabetes1,2, and lanifibranor—a nuclear-acting small-molecule triple agonist of PPARα, PPARγ and PPARδ—is in clinical phase 3 trials for the treatment of metabolic dysfunction-associated steatohepatitis3. Here, seeking to further improve the metabolic efficacy of GLP-1R–GIPR co-agonism, we report the development of a unimolecular quintuple agonist that combines the body weight-reducing and blood glucose-lowering effects of GLP-1R–GIPR co-agonism with the insulin-sensitizing and anti-inflammatory effects of lanifibranor via its targeted delivery into GLP-1R- and GIPR-expressing cells. In vitro, GLP-1–GIP–lanifibranor is indistinguishable from GLP-1–GIP in relation to incretin receptor signalling and shows equal stimulation of insulin secretion in isolated mouse islets. In vivo, however, GLP-1–GIP–lanifibranor outperforms GLP-1R–GIPR co-agonism and semaglutide, further decreasing body weight, food intake and hyperglycaemia in obese and insulin-resistant mice through synergistic incretin and PPAR action. The metabolic action of GLP-1–GIP–lanifibranor is blunted in mice with genetic or pharmacological inhibition of GLP-1R, GIPR or PPARδ and is absent in DIO double incretin receptor-knockout mice, collectively suggesting that GLP-1–GIP–lanifibranor has substantial therapeutic value in the treatment of obesity and diabetes.