2026-05-05 レンセラー工科大学(RPI)
AI-generated illustration showing a new way to treat chronic nerve pain by gently controlling a key protein instead of shutting it down completely
<関連情報>
- https://news.rpi.edu/2026/05/05/rpi-researchers-develop-new-approach-treating-chronic-neuropathic-pain-without-side
- https://pubs.acs.org/doi/10.1021/acs.jmedchem.5c03329
- https://www.nature.com/articles/s41467-026-69616-5
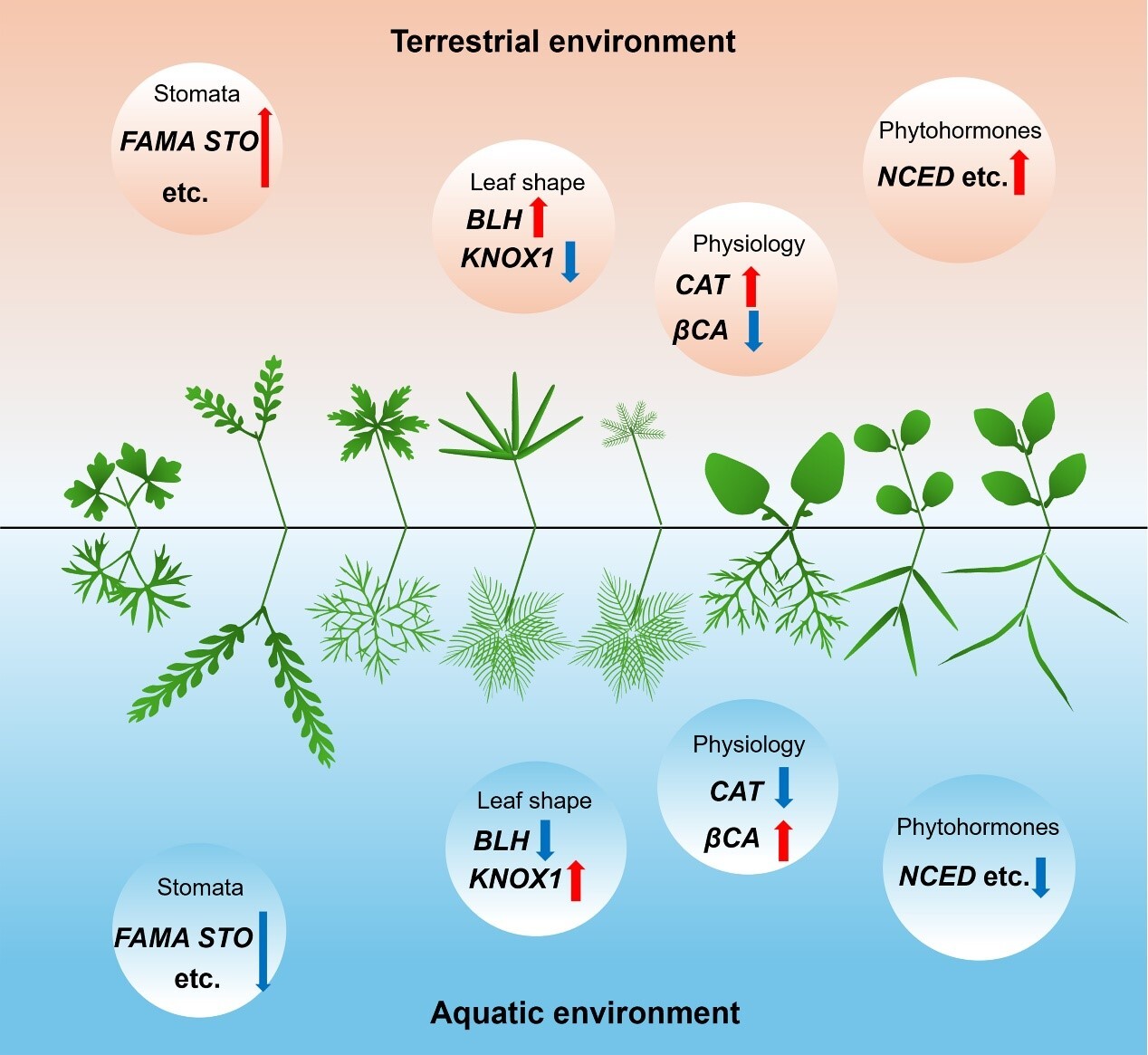
グリシントランスポーター2の非競合的かつ可逆的な阻害剤の同定:神経因性疼痛に対する有望な非オピオイド治療戦略 Identification of Non-competitive and Reversible Inhibitors of Glycine Transporter 2: A Promising Non-opioid Therapeutic Strategy for Neuropathic Pain
Christopher L. Cioffi,Tanmay K. Pati,Irina Lotsaris,Srinivasan Jayakumar,Julian Peiser-Oliver,Gaddam Mahesh,Ryan P. Cantwell Chater,Aravindan Jayaraman,Zachary J. Frangos,Krishna Suleria,Sally Evans,Ramasamy Kuppusamy,Arun Raja,Parthasarathy Muthuraman,Jay Mulmule,Suman Mishra,Snigdha Singh,Arunan Palanimuthu,Michael Michaelides,Azadeh Shahsavar,Sarasa Mohammadi,and Robert J. Vandenberg
Journal of Medical Chemistry Published: April 15, 2026
DOI:https://doi.org/10.1021/acs.jmedchem.5c03329
Abstract
Glycine transporter 2 (GlyT2) regulates extracellular glycine in the central nervous system (CNS) and is a promising target for restoring inhibitory neurotransmission in chronic pain. Although GlyT2 inhibitors show analgesic efficacy in rodent neuropathic pain models, the prototypical inhibitor ORG25543 (1) is limited by poor pharmacological properties, including slow dissociation kinetics and neuromotor toxicity at therapeutic doses, recapitulating the phenotype of homozygous GlyT2 knockout (GlyT2–/–) mice. To address these challenges, we developed a series of second-generation, conformationally restricted indoline analogues that act as noncompetitive and reversible GlyT2 inhibitors with varied potency and recovery kinetics. Among them, RPI-GLYT2-82 produced effective analgesia in murine neuropathic pain models without inducing excitatory neuromotor side effects observed with ORG25543. Collectively, these findings underscore the translational potential of reversible GlyT2 inhibitors as safe, nonopioid therapeutics for the management of neuropathic pain and provide a foundation for continued optimization of this chemotype toward preclinical candidate selection.
標的部位への副作用のない、神経因性疼痛に対する可逆的アロステリックGlyT2阻害剤 A reversible allosteric inhibitor of GlyT2 for neuropathic pain without on-target side effects
Ryan P. Cantwell Chater,Julian Peiser-Oliver,Tanmay K. Pati,Ada S. Quinn,Irina Lotsaris,Zachary J. Frangos,Kristen E. Anderson,Anna E. Tischer,Billy J. Williams-Noonan,Karin R. Aubrey,Megan L. O’Mara,Michael Michaelides,Sarasa A. Mohammadi,Christopher L. Cioffi,Robert J. Vandenberg & Azadeh Shahsavar
Nature Communications Published:16 February 2026
DOI:https://doi.org/10.1038/s41467-026-69616-5
Abstract
Chronic neuropathic pain, caused by nerve damage or disease, is increasing in prevalence, but current treatments are ineffective and over-reliant on opioids. The neuronal glycine transporter, GlyT2, regulates inhibitory glycinergic neurotransmission and represents a promising target for new analgesics. However, most GlyT2 inhibitors cause significant side effects, in part due to irreversible inhibition at analgesic doses. Here we develop a reversible inhibitor of GlyT2, RPI-GLYT2-82, and identify its binding site by determining cryo-EM structures of human GlyT2. We capture three fundamental conformational states of GlyT2 in the substrate-free state, and bound to either glycine, RPI-GLYT2-82 or the pseudo-irreversible inhibitor ORG25543. We demonstrate that RPI-GLYT2-82 dissociates from GlyT2 faster than ORG25543, providing analgesia in mouse neuropathic pain models without on-target side-effects or addiction liability. Our data provide a mechanistic understanding of allosteric inhibition of glycine transport, enabling structure-based design of non-opioid analgesics.