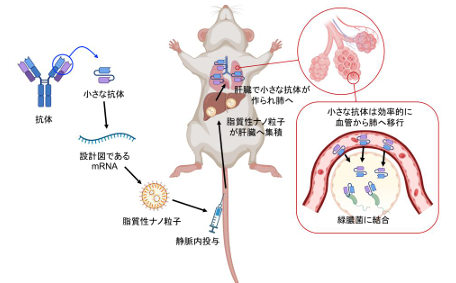
2026-05-02 京都大学iPS細胞研究所
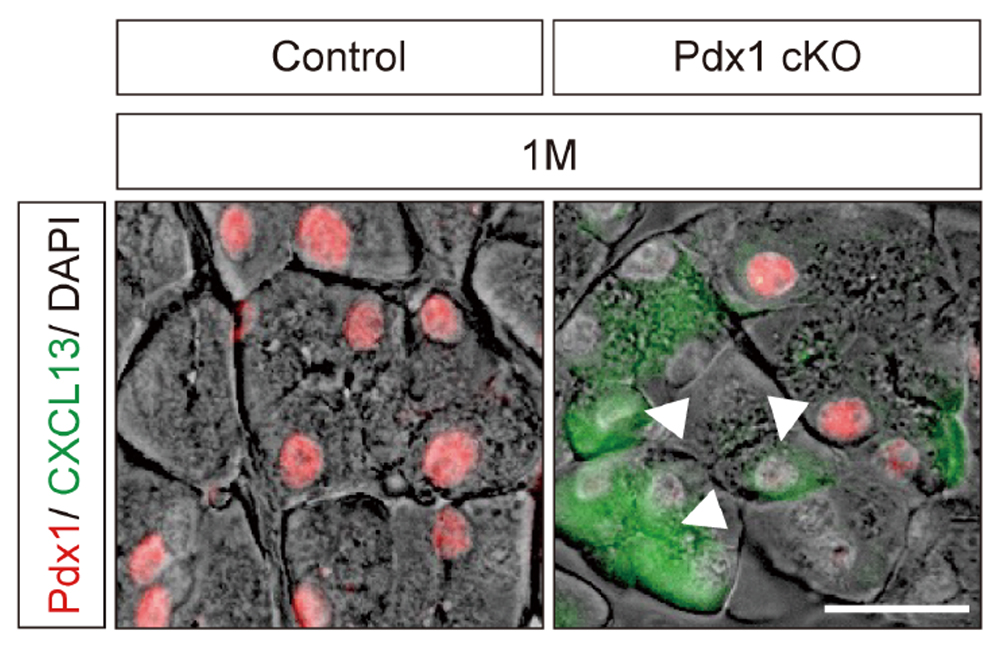
Fig.1 マウス膵臓の細胞を免疫染色した結果
<関連情報>
- https://www.cira.kyoto-u.ac.jp/j/pressrelease/news/260502-030000.html
- https://www.science.org/doi/10.1126/sciadv.adu7865
ストレスによって誘導されるCXCL13は、膵外分泌恒常性、加齢に伴う慢性炎症、および癌の進行を調節する Stress-induced CXCL13 regulates pancreatic exocrine homeostasis, age-related chronic inflammation, and cancer progression
Masahiro Yoshida, Kenichiro Furuyama, Keisuke Sumide, Masashi Horiguchi, […] , and Yoshiya Kawaguchi
Science Advances Published:1 May 2026
DOI:https://doi.org/10.1126/sciadv.adu7865
Abstract
Pancreatic cancers, whose incidence increases with age, are often refractory to treatment. Here, we identified a core mechanism shared by physiological homeostasis, senescent cell accumulation during aging, and pancreatic cancers. Pancreatic acinar cells, when stressed, secrete CXCL13, which protects stressed cells while transiently activating paracrine Hippo/YAP signaling to induce proliferation and PD-L1–mediated immune protection to maintain organ homeostasis. In the aged pancreas, CXCL13/YAP/PD-L1 signaling permits senescent cells to survive, driving feedforward chronic inflammation and steatosis. Because of prolonged CXCL13/YAP/PD-L1 activation in pancreatic cancers, neighboring noncancerous cells, activated for proliferation and immune-protected, eventually transform and accelerate tumor progression. CXCL13 blockade removed senescent cells and ameliorated steatosis in the aged pancreas while suppressing tumor growth in pancreatic cancer models, highlighting the CXCL13/YAP/PD-L1 axis as a potential therapeutic target. Together, our findings demonstrate the stress-induced CXCL13/YAP/PD-L1 axis as a central regulator of cell-state transitions in the pancreas, providing a unifying principle by which organ homeostasis, aging, and tumorigenesis are governed.