2026-03-17 東京科学大学
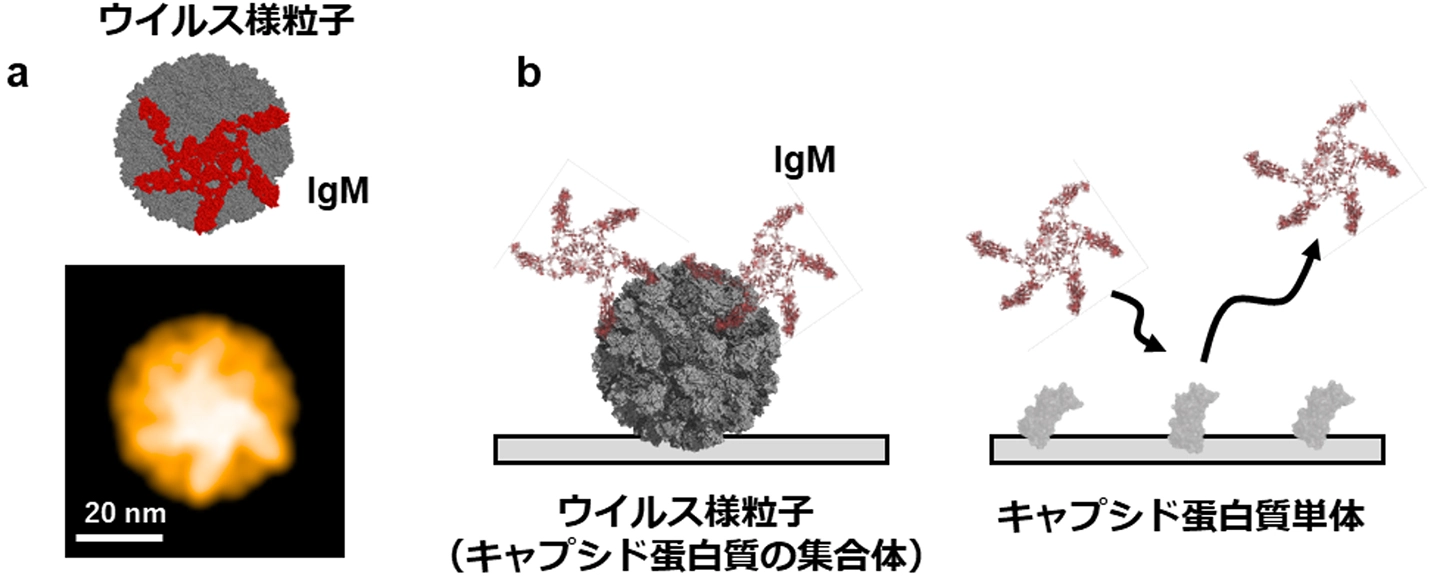
図1. ノロウイルスとIgMの相互作用
<関連情報>
- https://www.isct.ac.jp/ja/news/yzxme0kqokwc#top
- https://onlinelibrary.wiley.com/doi/10.1002/pro.70522
ノロウイルス変異株GII.4およびGII.17に対する高親和性IgMおよびIgGモノクローナル抗体の特性解析 Characterization of high affinity IgM and IgG monoclonal antibodies against norovirus variants GII.4 and GII.17
Jumpei Tagawa, Saeko Yanaka, Yuri Kato, Akitsu Masuda, Jae Man Lee, Akinobu Senoo, Kosuke Oyama, Takayuki Uchihashi, Motohiro Nishida, Takahiro Kusakabe, Jose M. M. Caaveiro
Protein Science Published: 12 March 2026
DOI:https://doi.org/10.1002/pro.70522
Abstract
Human noroviruses are a leading cause of acute gastroenteritis worldwide, yet the molecular principles governing antibody recognition of their highly repetitive capsid remain poorly understood. Here, we immunized mice with virus-like particles (VLPs) from the pandemic GII.4 strain and the emergent GII.17 strain, generating monoclonal IgM and IgG antibodies via hybridoma technology. High-speed atomic force microscopy visualized IgM antibodies scanning and engaging multiple protruding (P) domains on intact VLPs. Surface plasmon resonance (SPR) analyses of engineered antibodies with identical Fab sequences but different valencies revealed that, unlike monovalent IgG, multivalent IgM exhibits dramatic affinity gains—up to 100-fold—as antigen density increases. This avidity-driven enhancement arises from the dense, repetitive P-domain architecture of the norovirus capsid, enabling IgM to achieve high functional affinity despite modest intrinsic Fab binding. Our findings define how antibody valency and epitope organization cooperate to boost viral recognition, offering a mechanistic framework for designing next-generation vaccines and antiviral antibodies that harness multivalent engagement.