2026-03-26 京都大学
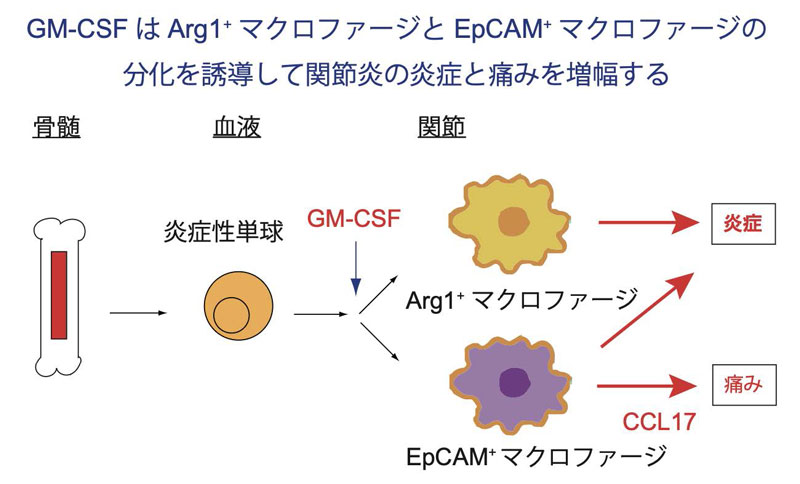
骨髄から血液中を移動してきた炎症性単球は関節に入るとGM-CSFの作用を受けてArg1⁺およびEpCAM⁺マクロファージに分化し関節の炎症増幅に関わる。特にEpCAM⁺マクロファージはCCL17を介して痛みにも関わっている。(作者:向山宙希)
<関連情報>
- https://www.kyoto-u.ac.jp/ja/research-news/2026-03-26
- https://www.kyoto-u.ac.jp/sites/default/files/2026-03/web_2603_Mukoyama-8050f7e9001882a8c2c1b9656ab03588.pdf
- https://www.science.org/doi/10.1126/sciadv.aec0986
GM-CSFは自己免疫性関節炎において炎症性マクロファージの機能的多様化をもたらす Pathogenic GM-CSF drives functional diversification of inflammatory macrophages in autoimmune arthritis
Hiroki Mukoyama, Yusuke Takeuchi, Daiya Ohara, Yoonha Lee, […] , and Keiji Hirota
Science Advances Published:25 Mar 2026
DOI:https://doi.org/10.1126/sciadv.aec0986
Abstract
Autoimmune T cells orchestrate joint inflammation and pain in concert with synovial macrophages; however, the mechanisms governing the development and functional diversification of these macrophages remain unclear. Using a model of T helper 17 cell (TH17 cell)–mediated autoimmune arthritis, we show that joint-infiltrating Ly6Chi monocytes in response to autoimmune TH17 cells, rather than resident synovial macrophages, are the primary mediators of disease pathogenesis. Granulocyte-macrophage colony-stimulating factor (GM-CSF), a critical component of the pathogenic circuit driven by arthritogenic TH17 cells, does not contribute to monocyte recruitment to the synovium but facilitates their subsequent differentiation into functionally distinct synovial macrophage subsets, thereby amplifying joint inflammation. Single-cell RNA sequencing identified two GM-CSF–dependent subpopulations of pathogenic synovial macrophages—Arginase-1+ and epithelial cell adhesion molecule (EpCAM)+ clusters—both expressing proinflammatory cytokines and matrix metalloproteinases. Notably, EpCAM+ macrophages uniquely express Ccl17, a pronociceptive mediator implicated in arthritic pain. Collectively, these findings delineate a GM-CSF–driven program of macrophage diversification that underpins both joint inflammation and pain, implicating this axis in the chronic activation of inflammatory and nociceptive pathways in autoimmune arthritis.