2026-04-01 東京科学大学
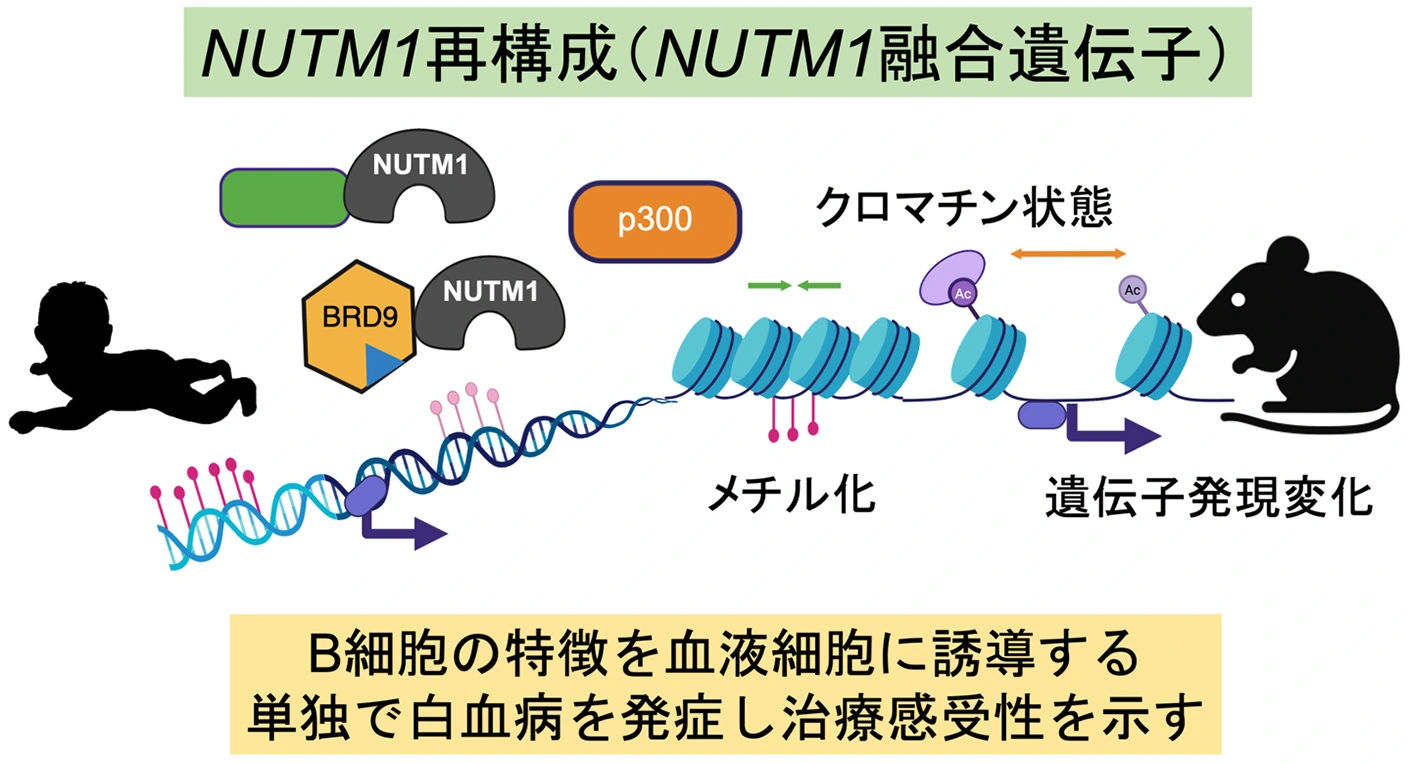
図1. NUTM1再構成乳児白血病の包括的解析(Created with BioRender.com)
<関連情報>
- https://www.isct.ac.jp/ja/news/vzx45fqdv6ln#top
- https://ashpublications.org/blood/article/147/13/1395/557529/Comprehensive-molecular-and-functional-analysis-of
NUTM1再構成白血病 の包括的な分子および機能解析 Comprehensive molecular and functional analysis of NUTM1-rearranged leukemia
Koutarou Nishimura,Tomoya Isobe,Tsukasa Shigehiro,Masaki Nomura,Weijia Zang,Muran Xiao,Wenjuan Liao,Yui Koike,Akira Nishimura,Aiko Sato-Otsubo,Hiromi Yamazaki,Hiromi Ito,Shinri Okada,Naomi Matsumoto,Wataru Saika,Yifan Zhang,Yumi Aoyama,Chihiro Hasegawa,Takaya Yamasaki,Yasuo Kubota,Kentaro Ohki,Nobutaka Kiyokawa,Genta Nagae,Kenichi Yoshida,Yasuhito Nannya,Hiroo Ueno,Shiro Fukuda,Kenji Tatsuno,Shuichi Tsutsumi,Yusuke Shiozawa,Yuichi Shiraishi,Kenichi Chiba,Hiroko Tanaka,Mariko Eguchi,Yuki Arakawa,Katsuyoshi Koh,Takao Deguchi,Daisuke Tomizawa,Takako Miyamura,Eiichi Ishii,Shuki Mizutani,Satoru Miyano,Hiroyuki Aburatani,Seishi Ogawa,Akifumi Takaori-Kondo,Akihiko Yokoyama,Omar Abdel-Wahab,Patricia Ernst,Junko Takita,Tomokatsu Ikawa,Masatoshi Takagi,Daichi Inoue
Blood Published:March 26, 2026
DOI:https://doi.org/10.1182/blood.2024026928
Key Points
- NUTM1 rearrangement in infant leukemia leads to unique transcriptomic and epigenetic landscapes, defining a distinct leukemia subtype.
- BRD9-NUTM1 drives transformation into B-lineage–committed ALL, while also enhancing sensitivity to chemotherapy.
Abstract
NUTM1 rearrangement defines a significant subset of B-cell acute lymphoblastic leukemia (B-ALL), particularly in infants lacking KMT2A rearrangements, yet its underlying molecular characteristics remain poorly understood. Here, we establish that NUTM1-rearranged (NUTM1-r) leukemia is a discrete entity characterized by a unique transcriptional and epigenetic landscape, notably featuring global DNA hypomethylation, irrespective of the 5′ fusion partner. Functional interrogation of NUTM1 fusions reveals a dual oncogenic role: they drive commitment toward the B-lymphoid lineage while concurrently conferring potent leukemic stem cell properties. Strikingly, expression of a representative fusion, BRD9-NUTM1, is sufficient to induce serially transplantable B-cell progenitor, prepro–B-like leukemia in vivo, faithfully recapitulating the key molecular and immunophenotypic features of human NUTM1-r B-ALL. Mechanistically, NUTM1 fusions establish an aberrant chromatin state, marked by global enhancement of H3K27 acetylation and the creation of distinctive open chromatin regions that co-opt both B-lineage and stemness-related transcriptional programs, including those involving NF-κB and posterior HoxA genes. In stark contrast to resistant KMT2A-rearranged leukemias, NUTM1-r leukemic cells exhibit a profound sensitivity to chemotherapy. This vulnerability is mechanistically linked to the leukemia’s dependence on active transcription. Our findings delineate the unique molecular profile of NUTM1-r leukemias, revealing specific vulnerabilities that rationalize their favorable clinical outcomes and suggest opportunities for modified therapeutic strategies.