2026-04-02 浙江大学(ZJU)
<関連情報>
- https://www.zju.edu.cn/english/2026/0402/c19573a3147180/page.htm
- https://www.cell.com/neuron/abstract/S0896-6273(25)01001-3
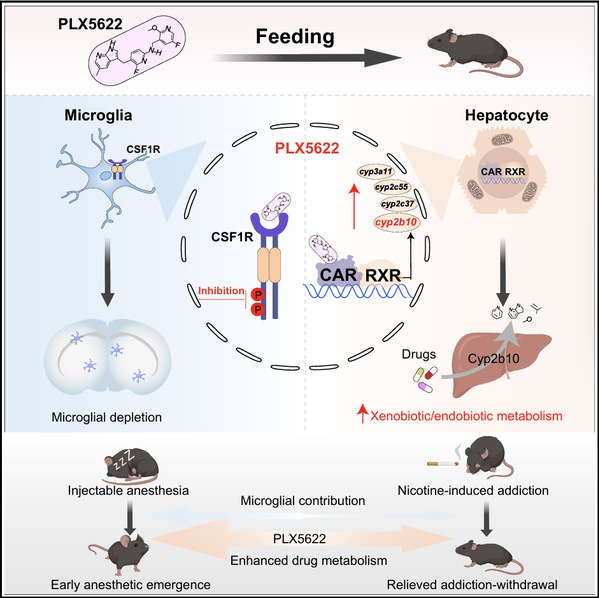
ミクログリアの減少にとどまらず、PLX5622は肝臓の構成的アンドロスタン受容体を活性化し、麻酔と依存性を変化させる More than microglial depletion: PLX5622 activates the hepatic constitutive androstane receptor to alter anesthesia and addiction
Kelei Cao (曹克磊) ∙ Wang Cheng (程旺) ∙ Liyao Qiu (邱礼耀) ∙ … ∙ Cunqi Ye (叶存奇) ∙ Shumin Duan (段树民) ∙ Zhihua Gao (高志华)
Neuron Published:February 23, 2026
DOI:https://doi.org/10.1016/j.neuron.2025.12.044
Highlights
- PLX5622 treatment shortens injectable, but not inhalation, anesthesia
- PLX5622 treatment induces xenobiotic responses in mice
- PLX5622 inhibits CSF1R but activates hepatic mouse CAR
- CAR deficiency prevents PLX5622-induced shortening of injectable anesthesia
Summary
The colony-stimulating factor 1 receptor (CSF1R) inhibitor PLX5622 has been widely used to deplete microglia for functional characterization and therapeutic support. Although diverse outcomes have been described after PLX5622 treatment, whether these phenotypes solely reflect microglial functions remains to be determined. Here, we show that transgenic microglial depletion did not mimic the accelerated anesthetic arousal or the alleviated nicotine addiction withdrawal symptoms observed after PLX5622 treatment in mice. We further identify that PLX5622 potently activates the mouse constitutive androstane receptor (CAR), leading to prominent induction of hepatic enzymes. The induced enzymatic activity enhances the metabolism and clearance of anesthetics and nicotine, thereby contributing to anesthetic insensitivity and addiction relief. Inactivation of CAR abolished these effects of PLX5622, indicating that the impact of PLX5622 treatment cannot be attributed exclusively to microglial depletion. Our findings raise awareness in evaluating consequences of PLX5622 treatment and provide insights into the design of specific CSF1R inhibitors.


