2026-04-16 京都大学
<関連情報>
- https://www.kyoto-u.ac.jp/ja/research-news/2026-04-16-0
- https://www.kyoto-u.ac.jp/sites/default/files/2026-04/web_2604_Arai-ee5bdb66fd688f4e47af2c00641bfec6.pdf
- https://www.sciencedirect.com/science/article/pii/S2666636726002599
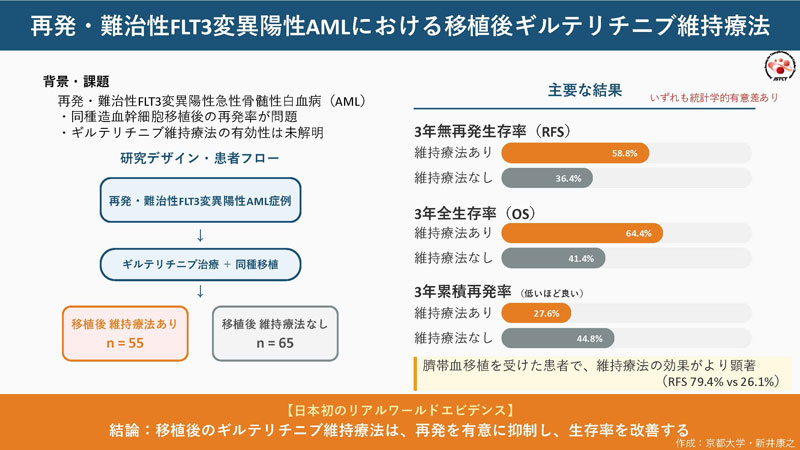
ギルテリチニブ維持療法は再発・難治性FLT3変異陽性急性骨髄性白血病の移植後再発を抑制する Maintenance treatment with gilteritinib suppresses post-transplant relapse in relapse/refractory FLT3-mutated acute myeloid leukemia
Yasuyuki Arai, Marie Ohbiki, Mio Kurata, Shuichi Ota, Masatsugu Tanaka, Kazunori Imada, Takahiro Fukuda, Yuta Katayama, Noriko Doki, Yoshinobu Kanda, Mamiko Sakata-Yanagimoto, Koichi Onodera, Nobuhiro Hiramoto, Noboru Asada, Takashi Ishida, Hiroki Uchida, Satoshi Uno, Takanori Teshima, Akiyoshi Takami, Takaaki Konuma …Shingo Yano
Transplantation and Cellular Therapy Available online: 12 April 2026
DOI:https://doi.org/10.1016/j.jtct.2026.03.042
Key points
- Dose adjustment may be necessary in post-HSCT gilteritinib maintenance therapy.
- Gilteritinib can be a beneficial maintenance treatment for R/R FLT3-mutated AML after HSCT.
Abstract
The FLT3 inhibitor, gilteritinib, has been widely used in patients with relapsed or refractory (R/R) FLT3-mutated acute myeloid leukemia (AML), but its tolerability and effectiveness after allogeneic hematopoietic stem cell transplantation (HSCT) are not well known outside of clinical trials.Therefore, we performed nation-wide surveillance in Japan. We used data from the national registry for patients transplanted including R/R FLT3-mutated AML patients treated with gilteritinib before and/or after HSCT. Post-HSCT administration dose and safety were summarized, and outcomes, including relapse-free survival (RFS) were investigated. We also used the historical cohort of HSCT before the approval of gilteritinib. Among R/R FLT3-mutated AML patients, 120 were treated with gilteritinib before and/or after allo-HSCT, and maintenance treatment was performed in 55 patients. The median initiation day was Day 47 after allo-HSCT (range 23–379) with a median starting dose of 80 mg (range 40–120 mg), and serious adverse events leading to gilteritinib dose reduction or temporary discontinuation were observed in 52.7% of these cases. The 3-year RFS was 46.8% and patients treated with post-HSCT gilteritinib had significantly better RFS (58.8%) than those without it (36.4%) (p < 0.001). Survival data in the historical cohort were similar to that of those who did not resume gilteritinib. In the subgroup analysis, post-HSCT gilteritinib showed an RFS benefit in cord blood transplantation (CBT; 79.4% vs. 26.1%, p < 0.001), but not in bone marrow or peripheral blood stem cell transplantation. This Japanese real-world study supports the tolerability and effectiveness of post-HSCT gilteritinib in R/R FLT3-mutated AML.