2026-04-16 ロックフェラー大学
<関連情報>
- https://www.rockefeller.edu/news/39382-therapeutic-proteins-antibodies-platform/
- https://www.science.org/doi/10.1126/science.adz8994
造血幹細胞の遺伝子編集によって作られたBリンパ球タンパク質工場 B lymphocyte protein factories produced by hematopoietic stem cell gene editing
Harald Hartweger, Chiara Ruprecht, Kai-Hui Yao, Philippe Laffont, […] , and Michel C. Nussenzweig
Science Published:16 Apr 2026
DOI:https://doi.org/10.1126/science.adz8994
Editor’s summary
Engineered B cells have the potential to deliver therapeutic antibodies or other proteins within the body. Hartweger et al. modified hematopoietic stem and progenitor cells (HSPCs) with genes encoding specific antibodies (see the Perspective by James and Zhang). When transferred into mice, the HSPCs developed into B cells that responded to vaccination and produced therapeutic quantities of antibodies. The B cells underwent some somatic hypermutation, a process that diversifies the antibodies that they produce. However, transferring two populations of HSPCs, each modified to make a different antibody, provided an alternative solution for delivery of broadly neutralizing antibodies. HSPCs could be modified so that they differentiated into B cells that produced a fluorescent protein alongside an antibody, suggesting that this approach may also be leveraged to deliver other types of protein therapeutics. — Sarah H. Ross
Structured Abstract
INTRODUCTION
Vaccines induce long-lasting expression of specific antibodies to control, eliminate, or prevent infections. In some cases, however, such as for HIV, influenza, or malaria, it has remained challenging to elicit effective, long-lasting antibody responses. Nevertheless, exceptional humans can produce broadly neutralizing antibodies (bNAbs) that can protect and treat infection when administered passively to others. Although eliciting bNAbs by vaccination remains an imperative, there remain multiple hurdles that may or may not be overcome. These include difficult-to-access epitopes that require exceptional levels of somatic mutation, the relative scarcity of bNAb-producing precursor cells, antibody-mediated masking effects, and requirements for substantial somatic hypermutation to create bNAbs.
RATIONALE
Rare hematopoietic stem cells (HSCs) are the source of all blood cells, including B lymphocytes. Upon vaccination, a small subset of antigen-specific B cells among the large polyclonal pool of B cells is selected and clonally expanded, and differentiates into plasma cells that produce high levels of long-lasting serum antibodies. To examine whether this self-amplifying system could be co-opted to produce a well-characterized protective or therapeutic antibody or an additional cargo protein, we developed a method to modify hematopoietic stem and precursor cells (HSPCs) that give rise to B lymphocytes. To determine whether B lymphocytes developing from such precursors can be activated to produce high titers of long-lasting antibodies, we exposed them to their cognate antigens by vaccination.
RESULTS
The immunoglobulin locus of HSPCs could be edited by CRISPR-Cas9 to encode specific antibodies and additional cargo proteins. When transplanted into mice, gene-edited HSPCs matured into B lymphocytes that expressed the antibody and cargo of interest before immunization. Exposure of mice to the cognate antigen by immunization induced long-lasting, high-serum titers of the inserted antibody, which could be increased by booster immunization. Very small numbers of edited HSPCs (~7000) were sufficient to achieve therapeutic levels of antibodies. Moreover, combinations of edited HSCs could be combined to produce different antibodies targeting the same immunogen simultaneously. The resulting titers were sufficient to neutralize HIV-1, inhibit Plasmodium falciparum traversal of hepatocytes, and protect mice from heterologous lethal influenza infection. Lastly, edited human HSPCs produced human B lymphocytes expressing the specific antibody in an immunodeficient mouse model.
CONCLUSION
Small numbers of edited HSPCs can develop into B lymphocytes that are clonally expanded by antigen to produce high titers of long-lasting protective or therapeutic antibodies and/or cargo proteins. Subject to further advances in delivery and safety, the system holds future therapeutic potential.
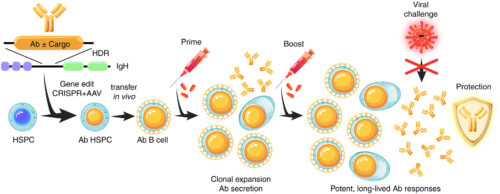
A prototype cell therapy approach.
Long-term, boostable expression of an antibody (Ab) and an additional cargo protein through hematopoietic stem cell gene editing. Expression in B lineage cells and amplification by immunization can mediate broad protection from lethal viral infection. AAV, adeno-associated virus; HDR, homology-directed repair; IgH, immunoglobulin heavy chain locus.
Abstract
Long-term in vivo production of therapeutic proteins and development of vaccines that elicit protective levels of broadly neutralizing antibodies (bNAbs) against major pathogens face challenges. In this study, we report on an alternative gene editing approach using small numbers of hematopoietic stem and progenitor cells (HSPCs) to direct long-term, high-level expression of antibodies or cargo proteins. In mice, edited B lymphocytes derived from transplanted HSPCs were activated by cognate antigen, underwent clonal expansion, and developed into specific antibody-synthesizing or cargo protein–synthesizing plasma cells. These cells produced long-lasting, therapeutic levels of serum antibody against HIV-1, malaria, or an anti-influenza virus bNAb that mediated universal protection from heterologous lethal challenge. Our data provide a paradigm for cell therapy approaches to prevent or treat disease using self-amplifying B cell protein factories.