2026-04-01 中国科学院(CAS)
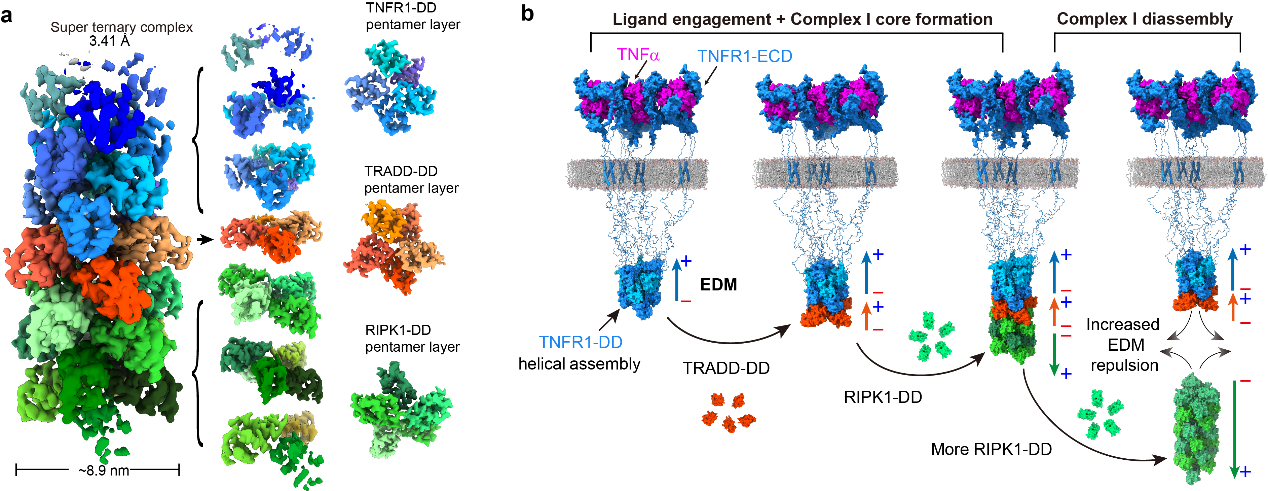
Mechanism for the dynamic assembly and disassembly of TNFR1 signaling Complex-I. (a) A helical super-complex consists of death domains of TNFR1, TRADD and RIPK1. (b) The force between electric dipole moments drives the dynamics of Complex-I. (Image by LIU Jianping)
<関連情報>
- https://english.cas.cn/newsroom/research-news/202604/t20260401_1154981.shtml
- https://www.nature.com/articles/s41586-026-10304-1
電気双極子モーメントがTNFR1複合体Iシグナロソームのダイナミクスを駆動する Electric dipole moment drives the dynamics of the TNFR1 complex I signalosome
Jianping Liu (刘建平),Jing Zhao (赵静),Jiayang Gao (高嘉阳),Kun Zhao,Yaoyao Han,Jing Yang,Zefei Li,Jianyu Ye,Ziyu Sun,Fengyi Wang,Xinyi Liu,Zekai Li,Siyu Ji,Bo Liu,Cong Liu,Yixiao Zhang,Junying Yuan & James J. Chou
Nature Published:01 April 2026
DOI:https://doi.org/10.1038/s41586-026-10304-1
Abstract
Dynamic assembly of the complex I signalosome mediated by three death domain (DD)-containing proteins—TNFR1, TRADD and RIPK1—is key for transmitting extracellular TNF stimuli to intracellular NF-κB signalling in controlling ‘live or die’ cell fate1. This signalling hub features the rapid recruitment of TRADD and RIPK1 after engagement of TNFR1 by TNF for the formation of complex I, followed by timed disassembly for transition into downstream signalling complexes2,3, but the mechanism driving the dynamic reversibility of complex I remains unclear. Here we captured the assembly core of complex I and determined its cryo-electron microscopy structure, showing a pentameric fibre comprising 31 DDs, with a single layer of a TRADD-DD pentamer sandwiched between multiple layers of TNFR1-DD and RIPK1-DD homopentamers. Structural analysis revealed a strong opposing electric dipole moment (EDM) generated by RIPK1-DD oligomerization relative to that of TNFR1-DD and TRADD-DD. Structure-guided mutagenesis in TNFR1–TRADD–RIPK1 pentameric fibres altering the EDM without affecting DD oligomerization demonstrated the role and mechanism of EDM in driving the dynamic reversibility mediating the rapid assembly and disassembly of complex I. Our study demonstrates a role for long-range interactions mediated by protein EDMs in driving the assembly and disassembly of super-signalling complex I for promoting NF-κB signalling.