2026-04-20 バッファロー大学(UB)
<関連情報>
- https://www.buffalo.edu/news/releases/2026/04/stop-alzheimers-linked-fibrils-from-forming.html
- https://www.nature.com/articles/s41467-026-69244-z#MOESM3
相分離と線維化の分離は、生体分子凝縮体の活性を維持する Decoupling phase separation and fibrillization preserves activity of biomolecular condensates
Tharun Selvam Mahendran,Anurag Singh,Sukanya Srinivasan,Christian M. Jennings,Christian Neureuter,Bhargavi H. Gindra,Sapun H. Parekh & Priya R. Banerjee
Nature Communications Published:16 February 2026
DOI:https://doi.org/10.1038/s41467-026-69244-z
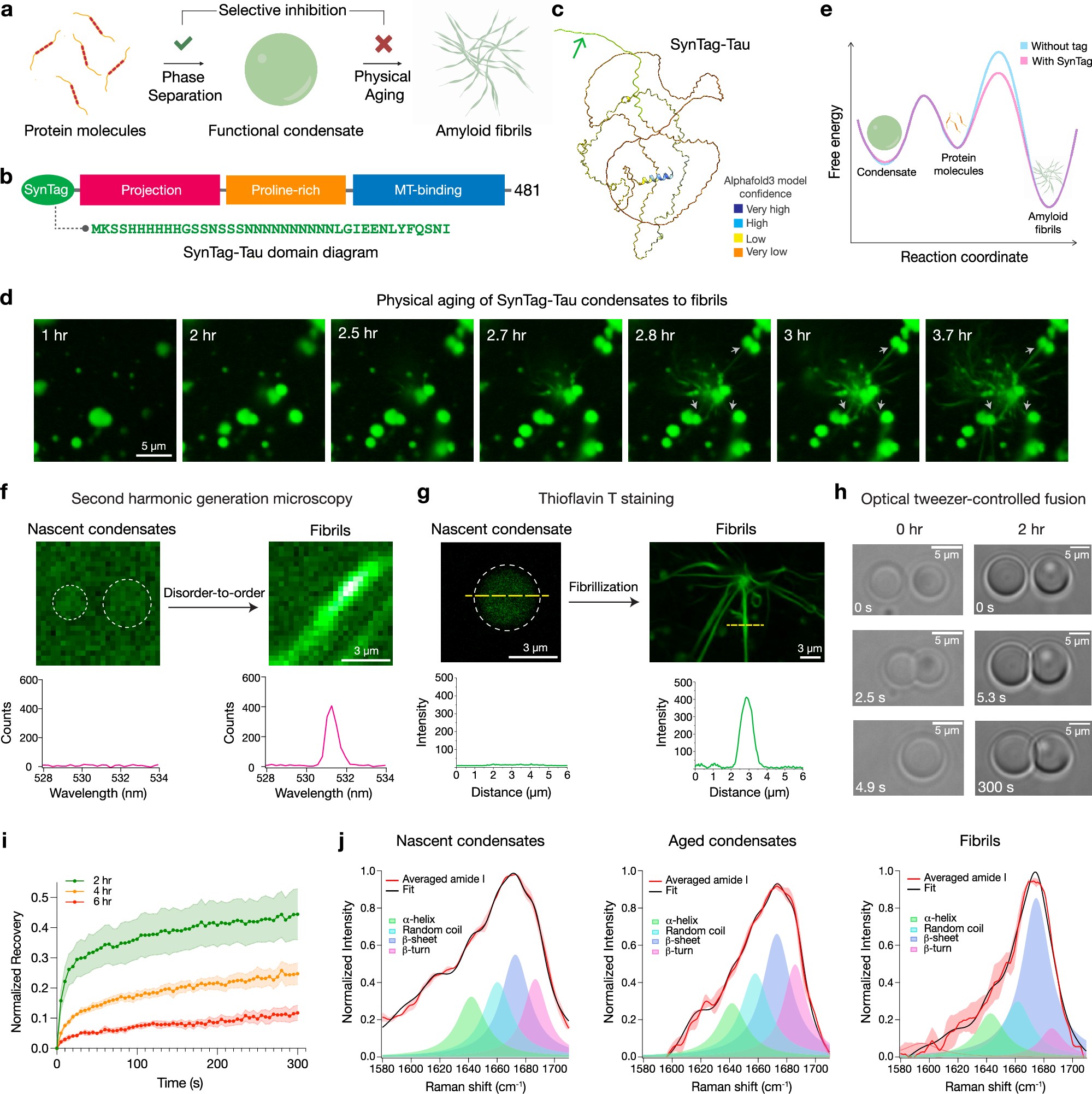
Abstract
The age-dependent transition of metastable, liquid-like protein condensates to amyloid fibrils is an emergent phenomenon in numerous neurodegeneration-linked protein systems. A key question is whether the thermodynamic driving forces underlying phase separation and maturation to amyloid fibrils are distinct and separable. Here, we address this question using an engineered version of microtubule-associated protein Tau, which forms biochemically-active condensates. These metastable protein condensates rapidly convert to amyloid fibrils under quiescent, cofactor-free conditions. In particular, the interfaces of condensates promote fibril nucleation, impairing condensate activity in recruiting tubulin and catalyzing microtubule assembly. Remarkably, a small molecule metabolite, L-arginine, selectively impedes age-dependent amyloid formation in a valence and chemistry-specific manner without perturbing phase separation. By enhancing condensate viscoelasticity, L-arginine counteracts the age-dependent decline in condensate activity. These results provide a proof-of-principle demonstration that small molecule metabolites can enhance the metastability of protein condensates and delay the formation of amyloid fibrils, thereby preserving biochemical function.

