2026-04-22 東京大学
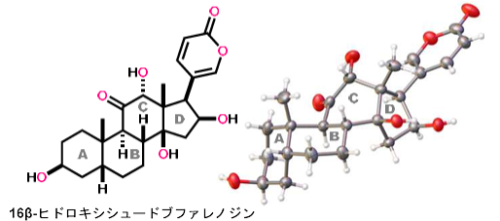
16β-ヒドロキシシュードブファレノジンの化学構造
<関連情報>
- https://www.u-tokyo.ac.jp/focus/ja/press/z0111_00062.html
- https://www.u-tokyo.ac.jp/content/400286434.pdf
- https://onlinelibrary.wiley.com/doi/10.1002/anie.5101280
16β-ヒドロキシルプソイドブファレノギンの収束型全合成 Convergent Total Synthesis of 16β-Hydroxylpseudobufarenogin
Wataru Shigematsu, Yo Matsumoto, Koichi Hagiwara, Masayuki Inoue
Angewandte Chemie International Edition Published: 20 April 2026
DOI:https://doi.org/10.1002/anie.5101280
ABSTRACT
16β-Hydroxylpseudobufarenogin (1), isolated from the venom of Bufo bufo gargarizans, has potent anticancer activity. The U-shaped steroidal structure of 1 possesses a cis-fused AB-ring system, a densely oxidized cis-fused CD-ring system, and a β-oriented 2-pyrone at C17. Herein, we present a new convergent strategy for assembling this complex steroidal architecture, culminating in the first total synthesis of 1 in 28 steps from (+)-Wieland–Miescher ketone. The AB- and D-ring fragments were coupled by Pd/Ag-promoted Suzuki–Miyaura coupling. Following the Co-catalyzed hydration of the D-ring, the C-ring was stereoselectively constructed by Ir-catalyzed radical-relay cyclization. Subsequent C-ring hydroxylation and installation of the β-oriented 2-pyrone through Pd/Cu-promoted Stille coupling and stereospecific epoxide rearrangement delivered 1. Because of its high chemo- and stereoselectivity, the present methodology would be applicable to the total synthesis of diverse oxygenated bufadienolides by simply altering the fragment structures.