2026-04-30 ロックフェラー大学
<関連情報>
- https://www.rockefeller.edu/news/39574-universal-blueprint-rna-rnap-gene-expression/
- https://www.cell.com/molecular-cell/fulltext/S1097-2765(26)00209-1
多サブユニット型DNA依存性RNAポリメラーゼ触媒活性の構造的基盤 Structural basis for multi-subunit DNA-dependent RNA polymerase catalytic activity
Andreas U. Mueller ∙ Seth A. Darst
Molecular Cell Published:April 30, 2026
DOI:https://doi.org/10.1016/j.molcel.2026.03.033

Highlights
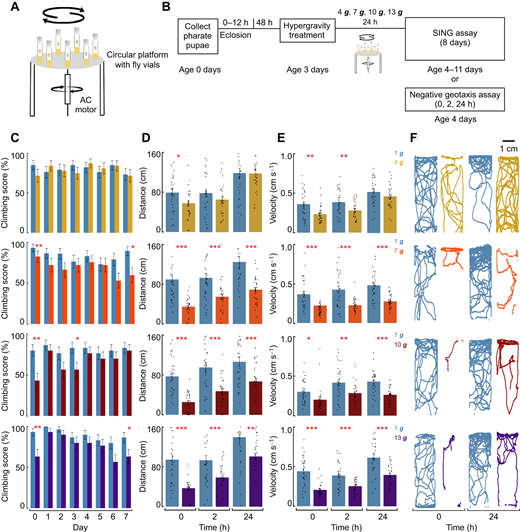
- Structures of bacterial RNA polymerase engaged in active RNA synthesis
- Native-substrate (Michaelis) and post-catalytic product complexes visualized
- Nucleotidyl transfer is catalyzed by specific acid-base chemistry
- A conserved water network in the active site suggests a proton wire
Summary
Multi-subunit DNA-dependent RNA polymerase (RNAP) is the central enzyme of transcription, yet its catalytic mechanism remains obscure because high-resolution structures of intermediates with native substrates are not available. We visualized E. coli RNAP on a promoter DNA template with nucleoside triphosphate substrates engaged in active RNA synthesis by cryo-electron microscopy. From this heterogeneous mixture, we determined five high-resolution structures of initial transcribing complexes, including a true Michaelis complex (MC) and a post-catalytic product complex (PC). The MC reveals key conformational transitions during catalysis. Waters in the MC and PC structures show striking overlap with those in corresponding S. cerevisiae RNA polymerase II (RNAPII) structures (see related paper by Li et al.), pointing to functional importance. Together, these results establish that RNAP catalyzes nucleotidyl transfer through a positional (entropic) mechanism, revealing structural determinants of the first step of gene expression.