2026-03-06 バージニア工科大学(VirginiaTech)
<関連情報>
- https://news.vt.edu/articles/2026/02/research-fralinbiomed-cancerrelapse.html
- https://www.nature.com/articles/s41409-025-02757-1
急性骨髄性白血病に対する同種移植前の測定可能な残存変異NPM1
Measurable residual mutated NPM1 before allogeneic transplant for acute myeloid leukemia
Rasha W. Al-Ali,Gege Gui,Niveditha Ravindra,Georgia Andrew,Devdeep Mukherjee,Zoë C. Wong,Ying Huang,Jason Gerhold,Matt Holman,Austin Jacobsen,Julian D’Angelo,Jeffrey Miller,Karina Elias,Jeffery J. Auletta,Firas El Chaer,Steven M. Devine,Antonio Martin Jimenez Jimenez,Marcos J. G. De Lima,Mark R. Litzow,Partow Kebriaei,Wael Saber,Stephen R. Spellman,Scott L. Zeger,Kristin M. Page,… Christopher S. Hourigan
Bone Marrow Transplantation Published:24 November 2025
DOI:https://doi.org/10.1038/s41409-025-02757-1
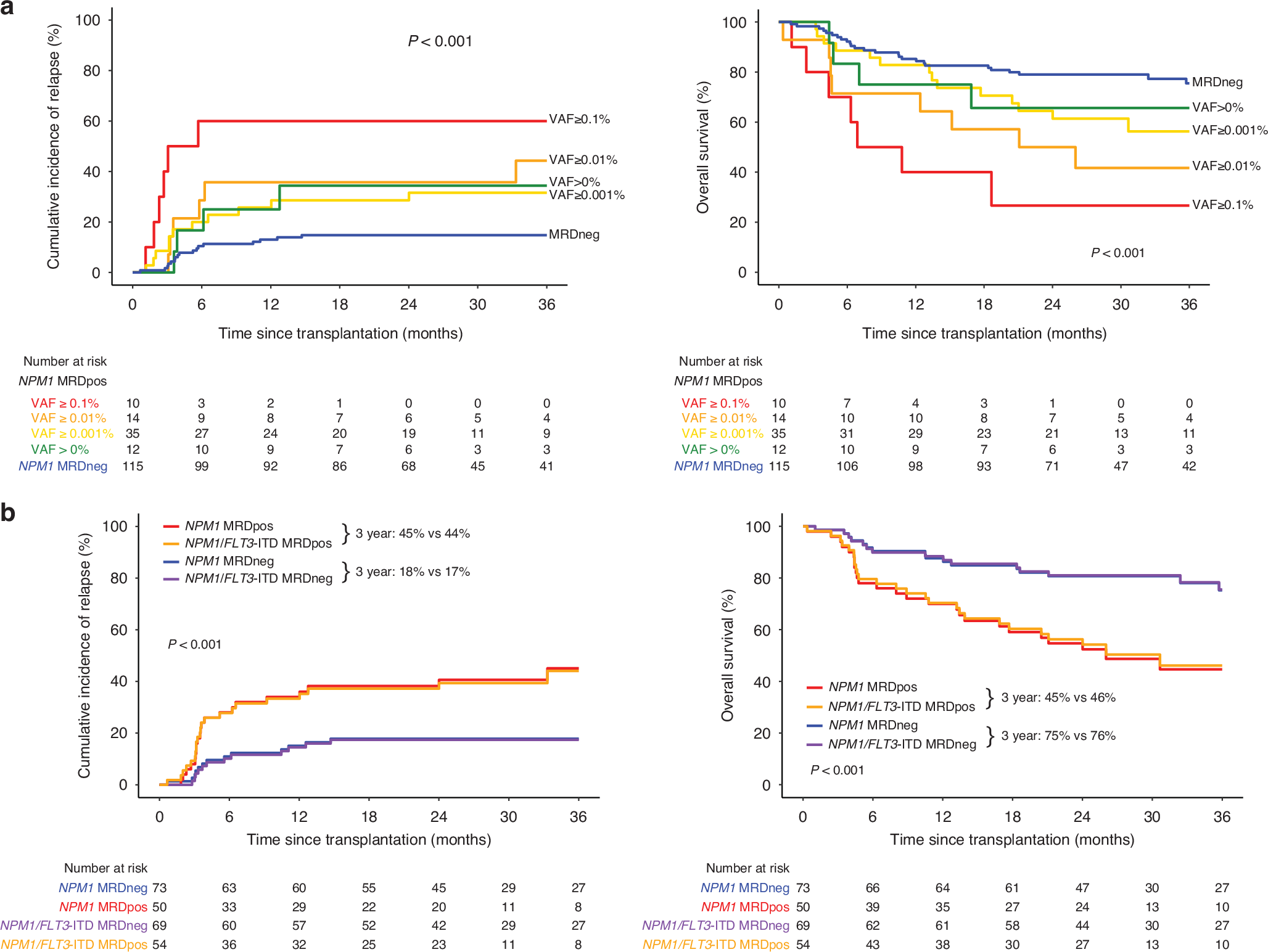
NPM1 mutations (NPM1m), seen in ~30% of adults with acute myeloid leukemia (AML), often co-occur with FLT3 internal tandem duplication (FLT3-ITD) [1, 2]. Measurable residual disease (MRD) testing in first complete remission (CR) identifies patients at high risk of relapse after allogeneic hematopoietic cell transplantation (alloHCT) [3, 4]. Quantitative RT-PCR assays are currently recommended for NPM1 MRD testing, while FLT3-ITD MRD testing is DNA-based using next generation sequencing (NGS) [5,6,7]. DNA-based NGS assays for NPM1 MRD testing may offer advantages over RNA, including improved template stability, accurate quantification, and high throughput capabilities, but require validation. We performed NPM1 MRD detection in blood collected from patients with AML during first CR (CR1) prior to alloHCT using a highly sensitive, commercially available DNA-based NPM1 NGS MRD assay.