2026-04-09 国立遺伝学研究所
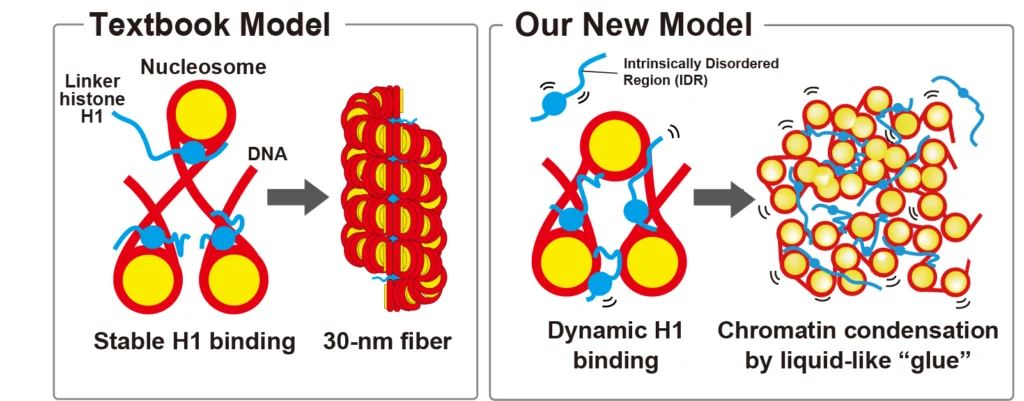
これまで、H1はヌクレオソームに安定に結合し、規則的な「かたい」構造を作ってクロマチンを凝縮させると考えられていた(図左)。今回の研究で、H1はダイナミックにヌクレオソームに結合し、「液体のり」のように働いてクロマチンを凝縮させ、不規則な「やわらかい」構造を作ることがわかった(図右)
<関連情報>
- https://www.nig.ac.jp/highlights/12279/
- https://www.nig.ac.jp/wp/wp-content/uploads/2026/04/PR20260409.pdf
- https://www.science.org/doi/10.1126/sciadv.aec9801
ヒト生細胞においてリンカーヒストンH1はクロマチンの「液体のり」として働く Linker histone H1 functions as a liquid-like glue to organize chromatin in living human cells
Masa A. Shimazoe, Jan Huertas, Charles Phillips, Satoru Ide, […] , and Kazuhiro Maeshima
Science Advances Published:8 Apr 2026
DOI:https://doi.org/10.1126/sciadv.aec9801
Abstract
Linker histone H1, the most abundant chromatin protein, condenses chromatin, modulates DNA transactions such as transcription and DNA replication/repair, and participates in differentiation, development, and tumorigenesis. While recent studies indicate that nucleosomes are clustered as condensed chromatin domains in higher eukaryotic cells, how histone H1 mechanically condenses chromatin remains unclear. Here, using a combination of direct visualization of single-H1 molecules in living human cells and multiscale molecular dynamics simulations, we demonstrate that the majority of H1 behaves like a liquid inside chromatin domains, rather than binding stably to nucleosomes as suggested by the traditional model. H1 functions as a liquid-like “glue,” mediating dynamic multivalent electrostatic interactions between nucleosomes within chromatin domains. Consistently, rapid depletion of H1.2 leads to decondensed chromatin domains both in cells and in silico. Our findings suggest that the H1 “glue” condenses chromatin domains while keeping them fluid and accessible, thereby supporting essential DNA transactions.