2026-04-09 清華大学
<関連情報>
- https://www.tsinghua.edu.cn/en/info/1245/14763.htm
- https://www.science.org/doi/10.1126/science.adw1803
脳室下帯の放射状グリア細胞は、ヒトの脳における抑制性ニューロンの産生を維持する Subventricular zone radial glial cells maintain inhibitory neuron production in the human brain
Longzhong Jia, Xiaohan Li, Yiming Yan, Linhe Xu, […] , and Da Mi
Science Published:15 Jan 2026
DOI:https://doi.org/10.1126/science.adw1803
Editor’s summary
The vast majority of cortical γ-aminobutyric acid–sensitive inhibitory neurons are generated in the subpallium, specifically the medial ganglionic eminence (MGE). Jia et al. characterized the cellular composition of the human MGE using a combination of single-cell RNA sequencing and spatial transcriptomic approaches across a wide range of fetal developmental stages (see the Perspective by Bonafina and Nguyen). They identified a progenitor cell type in the human MGE subventricular zone responsible for maintaining the generation of interneurons and glial cells throughout human brain development. The authors also used computational methods to explore the trajectories of MGE-derived neuronal populations and linked specific MGE cell types to neuronal populations. The results provide valuable insights into the mechanisms mediating human brain development. —Mattia Maroso
Structured Abstract
INTRODUCTION
The expansive human cerebral cortex, with its 16 billion neurons, is ranked the highest in the number of cortical neurons among all species. Such expansion is accompanied by an increase in the number of not only glutamatergic excitatory neurons and γ-aminobutyric acid–expressing but also GABAergic inhibitory neurons in evolutionary history. The greater number and diversity of GABAergic inhibitory neurons shape the signature properties of circuit assemblies in the human cerebral cortex, which potentially contribute to humans having the highest cognitive capacity among all species. However, how the human brain generates its vast repertoire of GABAergic inhibitory neuron remains unclear.
RATIONALE
Cortical outer radial glia (oRG, also known as bRG), a primate-enriched progenitor population in the cortical outer subventricular zone, has been identified as a key driver of the expanded production of excitatory neurons through prolonged neurogenesis. By contrast, most cortical GABAergic inhibitory neurons originate from the subpallium, specifically the medial ganglionic eminence (MGE), an important progenitor domain in the developing brain. Notably, the MGE in primates—particularly in the human species—shows increased complexity through the evolutionarily expanded subventricular zone (SVZ). This raises a pivotal question: Is there a subpallial progenitor population, akin to oRG cells, that amplifies inhibitory neuron production?
RESULTS
In this study, we combined spatial and single-cell transcriptomics to depict a comprehensive cellular and molecular landscape of human MGE (hMGE) development from gestational week (GW) 9 to 39. We identified spatiotemporally and molecularly segregated progenitor populations that were fated to produce distinct types of GABAergic and cholinergic inhibitory neurons, indicating that the emergence of neuronal diversity was linked to spatial and temporal specification of progenitor cells in the hMGE. Notably, we uncovered an evolutionarily distinct SVZ radial glial cell (SVZ RGC) population in the hMGE, which had homologous cell type in macaques but not in mice. The SVZ RGC exhibits distinct molecular features, spatiotemporal distribution, cellular morphology, and mitotic behavior. By reconstructing the developmental trajectory of hMGE cells, we demonstrated that SVZ RGCs maintain the production of GABAergic inhibitory neurons and glial cells throughout human brain development and thus provide a cellular basis for the extended production of human inhibitory neurons in an evolutionary context.
CONCLUSION
Our study reveals evolutionarily distinct features of human inhibitory neuron generation and shed light on the mechanisms underlying human brain development. Notably, the hMGE SVZ RGC is a new addition to the pantheon of neural progenitor cell types, marking an important cytogenic source in the human brain.
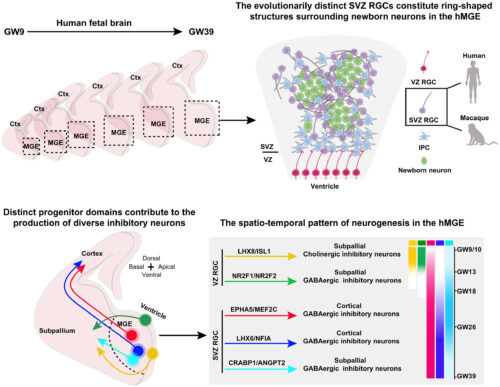
The evolutionarily distinct hMGE SVZ RGCs maintain the production of GABAergic inhibitory neurons in the developing human brain.
We depicted a comprehensive cellular and molecular landscape of hMGE development and uncovered an evolutionarily distinct RGC type with distinct molecular and cellular features. We identified spatiotemporally and molecularly segregated progenitor cell populations (color-coded progenitor domains) fated to produce distinct types of inhibitory neurons (color-coded arrows).
Abstract
The number and diversity of inhibitory neurons (INs) increased substantially during mammalian brain evolution. However, the generative mechanisms of the vast repertoire of human INs remain elusive. We performed spatial and single-cell transcriptomics of human medial ganglionic eminence (hMGE), a pivotal source of cortical and subpallial INs, and built the trajectories of hMGE-derived cells during brain development. We identified spatiotemporally and molecularly segregated progenitor cell populations fated to produce distinct IN types. We uncovered an evolutionarily distinct progenitor cell type in the hMGE subventricular zone that maintained the production of INs and glial cells throughout human brain development. Our findings reveal evolutionarily distinct features of IN generation and shed light on the distinct mechanisms underlying human brain development.