2026-03-31 ミュンヘン大学(LMU)
<関連情報>
- https://www.lmu.de/en/newsroom/news-overview/news/precision-work-prior-to-cell-division-how-enzymes-optimize-dna-structure-53b98b01.html
- https://www.nature.com/articles/s41467-026-70698-4
Dbf4依存性キナーゼは染色体複製起点におけるIno80の機能を微調整する Dbf4-dependent kinase finetunes Ino80 function at chromosome replication origins
Priyanka Bansal,Shibojyoti Lahiri,Chandni Natalia Kumar,Jessica Furtmeier,Lorenz Spechtenhauser,Lorenzo Galanti,Juan de Dios Barba Tena,Erika Chacin,Garp Linder,María Ángeles Ortíz-Bazán,Marisa Müller,Petra Vizjak,Tobias Straub,Felix Mueller-Planitz,Johannes Stigler,Andrés Aguilera,Belen Gómez-González,Boris Pfander,Philipp Korber,Axel Imhof & Christoph F. Kurat
Nature Communications Published:28 March 2026
DOI:https://doi.org/10.1038/s41467-026-70698-4
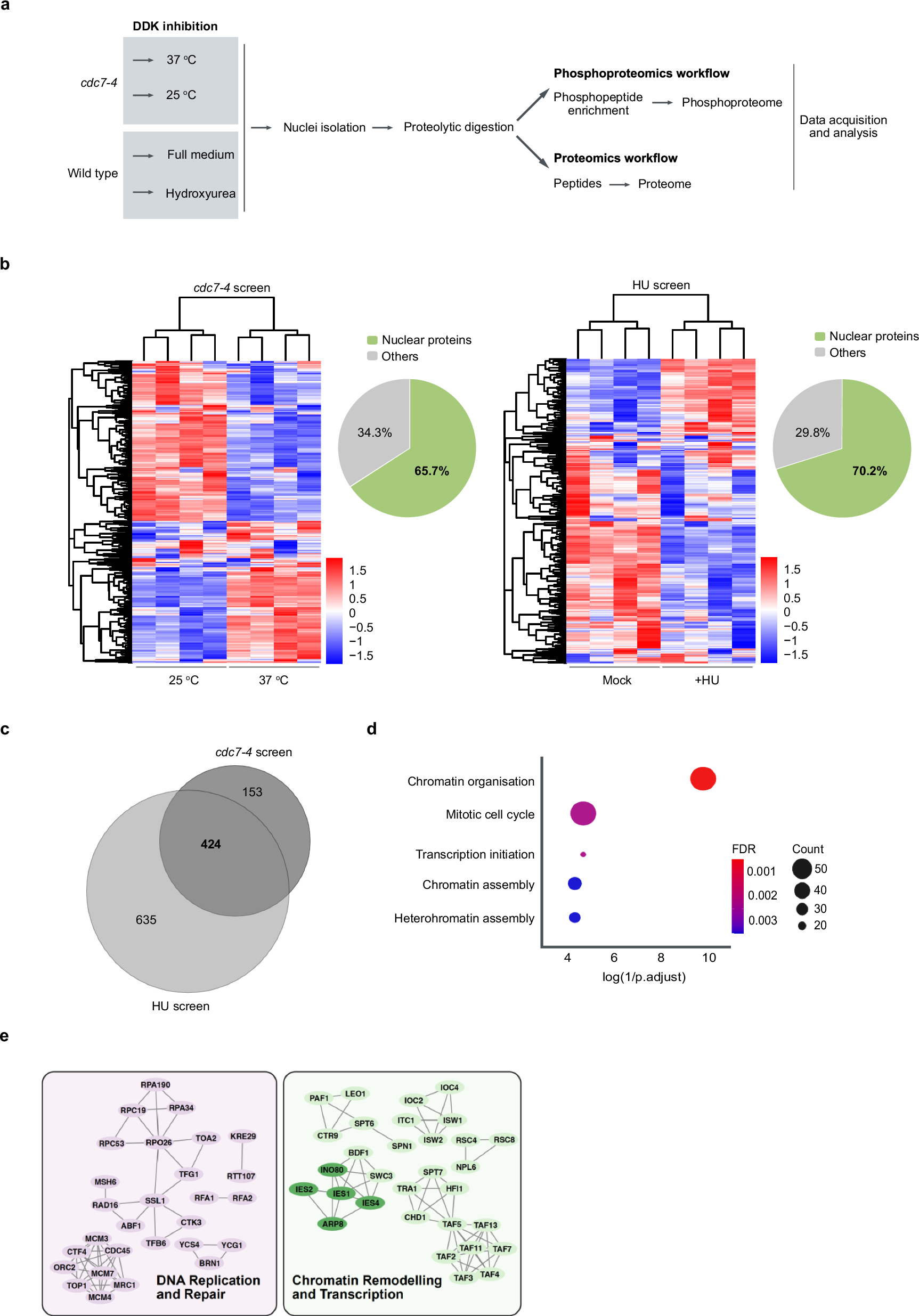
Abstract
The highly conserved Dbf4-Dependent Kinase (DDK) plays a pivotal role during S phase. It phosphorylates the replicative helicase (minichromosome maintenance, MCM complex), which leads to the initiation of replication. However, few other targets, besides the MCM complex, are known, leaving DDK an understudied kinase. Here, we determine the nuclear DDK-dependent phosphoproteome by a two-pronged mass spectrometry approach. Among ~ 400 DDK-dependent phosphorylation targets, we find the Arp8 subunit of the INO80 chromatin remodeling complex. Arp8 phosphorylation stabilises INO80’s complex integrity, finetunes its nucleosome spacing at replication origins, stimulates replication and improves the replication stress response. Taken together, we report the regulation of a chromatin remodeler with nucleosome-spacing activity by the cell-cycle machinery. DDK not only regulates the core replication machinery but also regulates a factor that generates replication-conducive chromatin architecture at replication origins.


