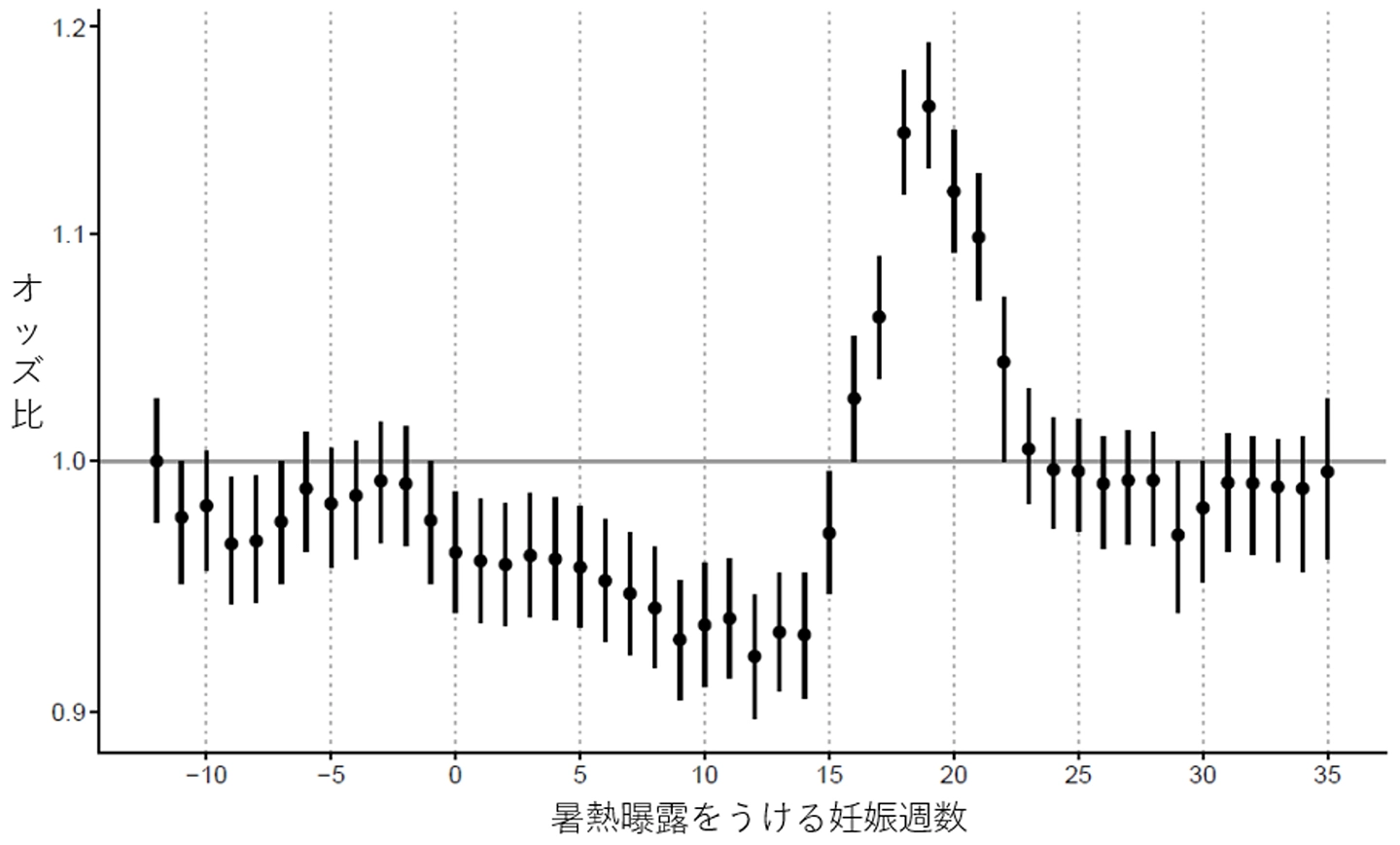
2026-04-23 東京科学大学
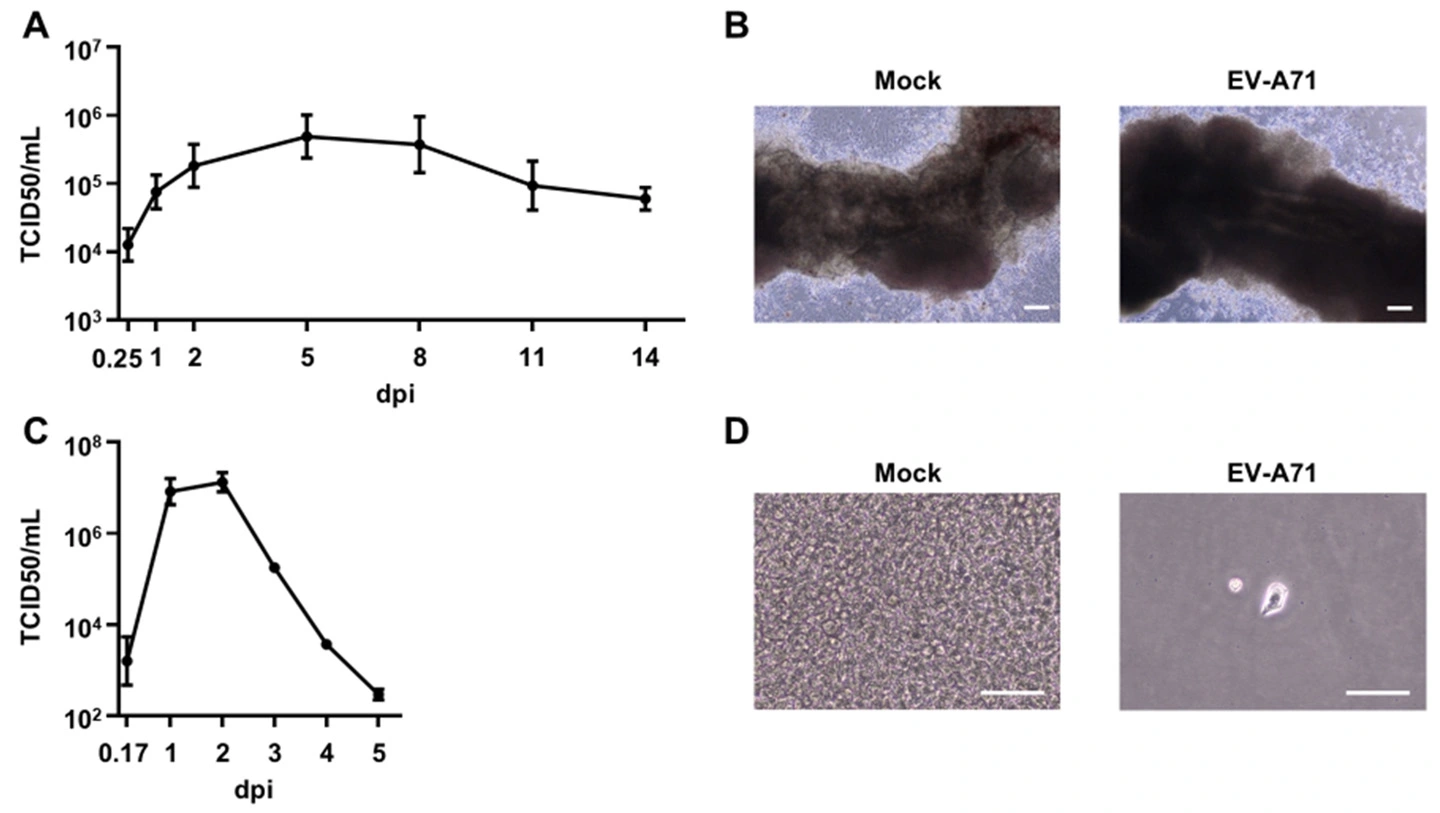
図1. A-B: 腸管MPSを用いたEV-A71感染実験。
A: 腸管MPSにEV-A71を感染させ、培養上清中のウイルス力価を測定した。
B: EV-A71感染後14日目における腸管MPSの位相差画像を示す。スケールバーは200 μm。
C-D: RD細胞を用いたEV-A71感染実験。
C: RD細胞にEV-A71を感染させ、培養上清中のウイルス力価を測定した。
D: EV-A71感染後4日目におけるRD細胞の位相差画像を示す。スケールバーは100 μm。
<関連情報>
腸管微生理システムを用いたヒトエンテロウイルスA71感染のモデル化 Modeling human enterovirus A71 infection using an intestinal microphysiological system
Hiroki Futatsusako, Sayaka Deguchi, Kaori Kosugi, Rina Hashimoto, Noriyo Nagata, Tadaki Suzuki, Takuya Yamamoto, Kazuo Takayama
Journal of Virology Published:21 April 2026
DOI:https://doi.org/10.1128/jvi.00250-26
ABSTRACT
Enterovirus A71 (EV-A71), a causative virus of hand, foot, and mouth disease, primarily infects and replicates in the intestine and, in severe cases, spreads to the central nervous system, leading to neurological complications. Therefore, suppressing viral replication in the intestine is important to prevent severe complications. However, the intestinal pathophysiological changes in EV-A71-infected patients remain poorly understood. In this study, we aimed to examine the intestinal response to EV-A71 infection using the intestinal microphysiological system (MPS) we previously developed using human pluripotent stem cells and microfluidic devices. The viral titers were detectable in the cell culture supernatant of the intestinal MPS for 14 days after the viral infection. Despite this, EV-A71 infection did not induce significant morphological changes in the intestinal MPS or alter the expression of epithelial cell markers, suggesting that the virus can infect the intestinal MPS without causing intestinal epithelial damage. In addition, we found that the secretion of interferons (IFNs) in the cell culture supernatant was not increased by viral infection. Interestingly, treatment with recombinant IFNs increased the expression of innate immune response-related genes and reduced viral mRNA levels. A strong association was observed between EV-A71 infection and IFN signaling in the intestinal MPS. We believe that the intestinal MPS would be a valuable platform for studying EV-A71 infection and evaluating antiviral strategies.