2026-04-28 カナダ・ブリティッシュコロンビア大学(UBC)
<関連情報>
- https://news.ubc.ca/2026/04/targeting-undruggable-cancer-proteins/
- https://www.nature.com/articles/s41392-026-02642-3
アンドロゲン受容体の内在的に無秩序な転写活性化ドメインを標的とした薬物療法 Drugging the intrinsically disordered transactivation domain of androgen receptor
Jon K. Obst,Carmen A. Banuelos,Kunzhong Jian,Amy H. Tien,Oleksandr A. Shkrabak,Jun Wang,Nasrin R. Mawji,Teresa Tam,Marija Vuckovic,David E. Williams,Jason C. Rogalski,Xiaojing Yuan,Natalie C. J. Strynadka,Raymond J. Andersen & Marianne D. Sadar
Signal Transduction and Targeted Therapy Published:28 April 2026
DOI:https://doi.org/10.1038/s41392-026-02642-3
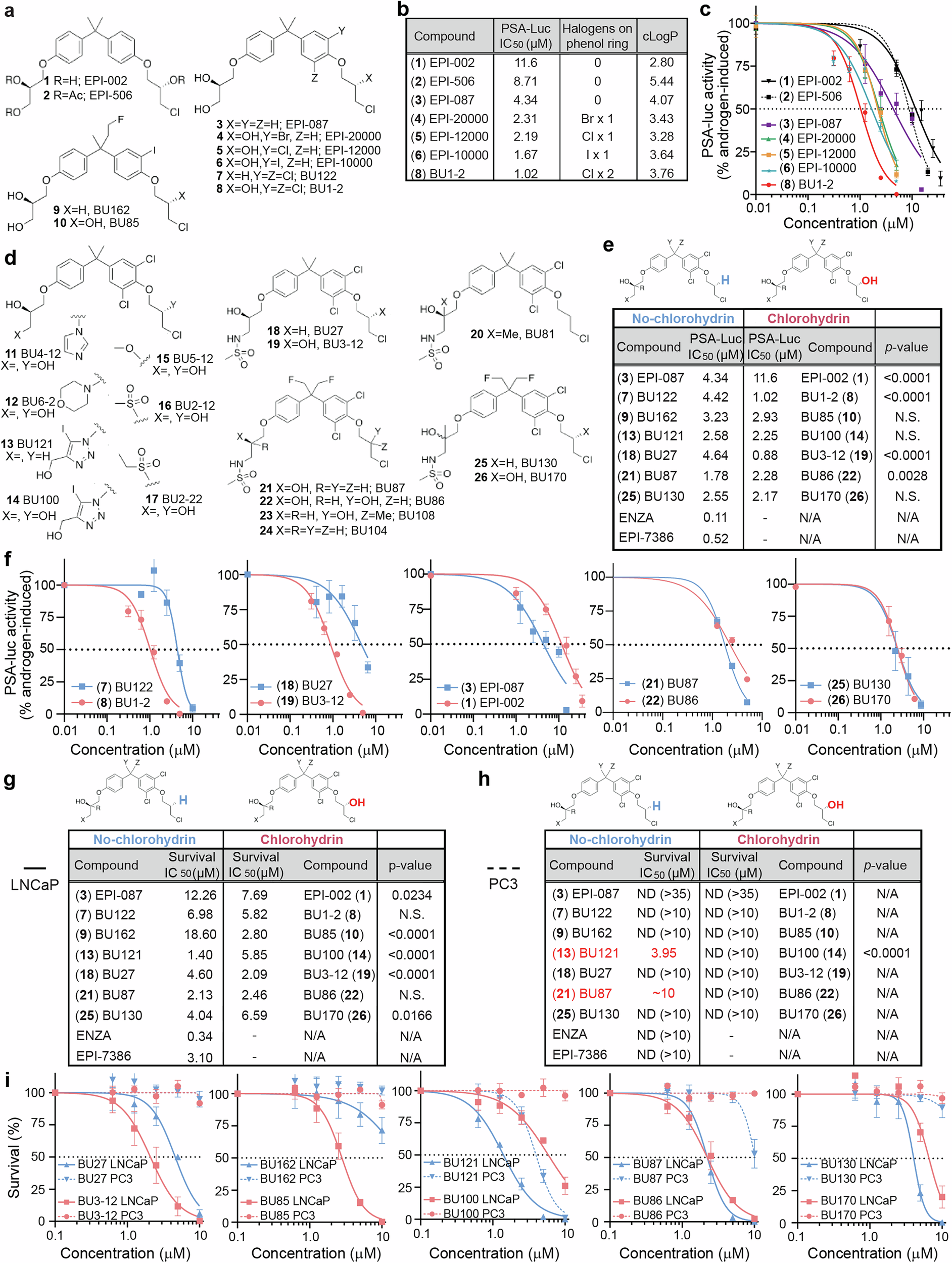
Abstract
Androgen receptor (AR) is a therapeutic target for prostate cancer. Despite effectively targeting its folded ligand-binding domain (LBD), resistance ultimately develops by mechanisms involving reactivation of AR signaling. These mechanisms include expression of constitutively active AR that lacks LBD and fueled the discovery of inhibitors that bind to AR’s N-terminal intrinsically disordered transactivation domain (TAD). AR-TAD inhibitors (ARTADIs) are unique due to the paucity of small molecule inhibitors that bind directly to intrinsically disordered TADs, which have historically been considered undruggable. Leveraging our library of ARTADIs using cultured prostate cancer cells and multiple xenograft models, we reveal that small alterations in the chemical scaffold impact selectivity and potency within the AR-transcriptome; impacting signal transduction pathways involved in protumorigenic mechanisms. Mechanistically, these compounds differentially disrupt interactions between full-length AR or splice-variant AR-V7, and co-regulators, as revealed by rapid immunoprecipitation mass spectrometry of endogenous protein and the proximity ligation assay. Biophysically, several ARTADIs displayed exceptionally strong binding affinities that were better than, or were comparable to the LBD-inhibitor enzalutamide, with dissociation constants in the picomolar to low-nanomolar range as determined by surface plasmon resonance and microscale thermophoresis. MS/MS analysis revealed covalent binding to cysteine 129. In vivo, ARTADIs outperformed enzalutamide against prostate cancer xenografts in the presence of androgens, underscoring the therapeutic potential of targeting alternative AR domains. These findings support the feasibility – but also highlight the complexity – of developing drugs against an intrinsically disordered TAD impacted by multivalent binding interactions that may not occur in a stepwise fashion.