2026-05-07 中国科学院(CAS)
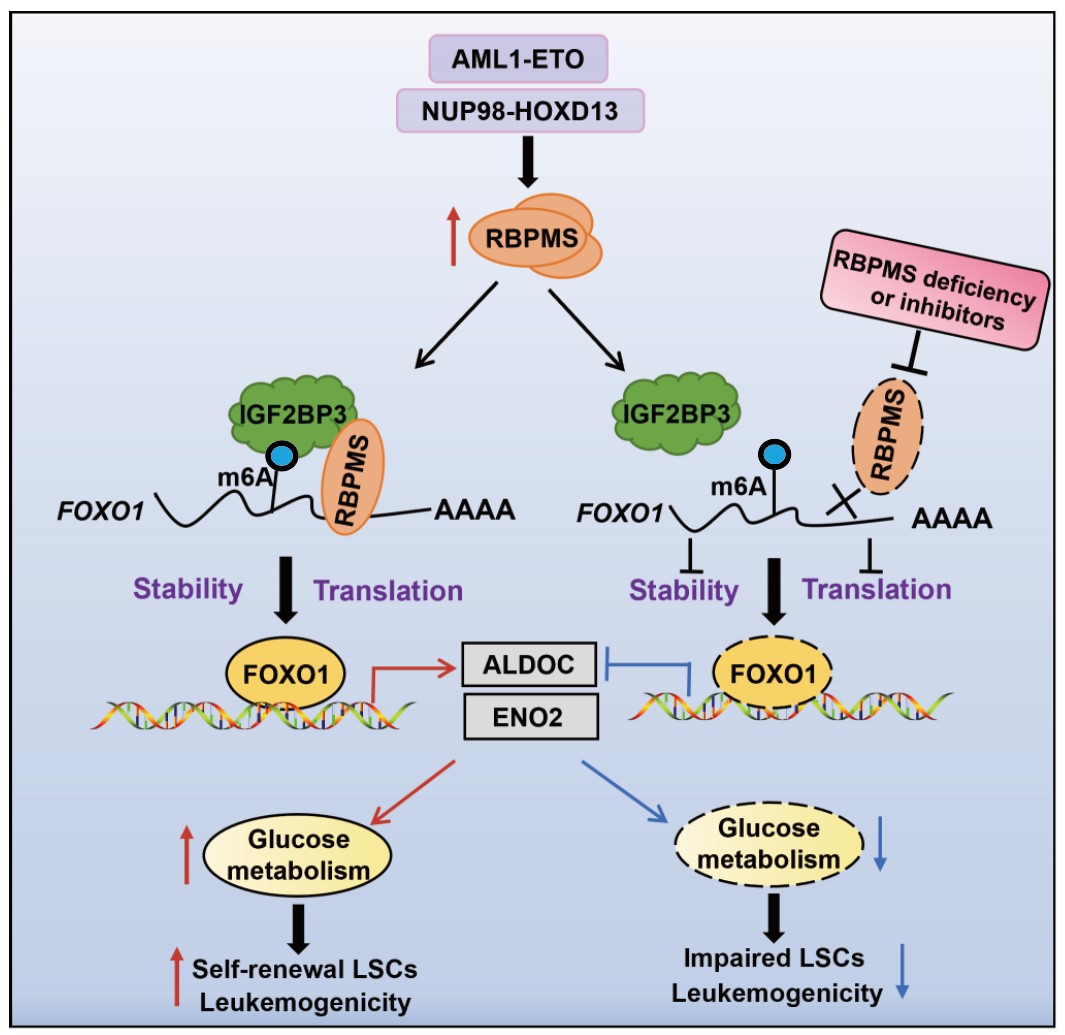
Molecular Mechanisms and Targeted Strategies for RBPMS Regulation of Acute Myeloid Leukemia Onset and Progression. (Image by Dr. WANG Lan’s team)
<関連情報>
- https://english.cas.cn/newsroom/research-news/202605/t20260506_1158335.shtml
- https://www.science.org/doi/10.1126/scitranslmed.adv8951
RBPMSを標的とすることで、急性骨髄性白血病のマウスモデルにおいてFOXO1を介した幹細胞シグネチャーを選択的に除去できる Targeting RBPMS selectively eliminates FOXO1-mediated stem cell signatures in mouse models of acute myeloid leukemia
Ping Liu, Sulin Zhang, Bing-Yi Chen, Binhe Chang, […] , and Lan Wang
Science Translational Medicine Published:6 May 2026
DOI:https://doi.org/10.1126/scitranslmed.adv8951
Editor’s summary
Leukemia is a cancer derived from blood and associated with poor patient prognosis, highlighting the need for new treatments. Here, Liu et al. evaluate targeting RNA binding protein with multiple splicing (RBPMS) to inhibit leukemogenesis. They show that RBPMS recruits IGF2BP3 to induce expression of FOXO1, which is involved in glucose metabolism and self-renewal of leukemic stem cells (LSCs). They show that inhibition of RBPMS delayed leukemia progression in acute myeloid leukemia (AML) patient-derived xenograft (PDX) models, offering a promising target that warrants further exploration. —Dorothy Hallberg
Abstract
Leukemia is a malignant tumor with a high recurrence rate and poor prognosis for patients. Thus, there is an urgent need to explore new therapeutic targets that play critical roles in leukemogenesis but have little effect on normal hematopoietic cells. Here, we show that RNA binding protein with multiple splicing (RBPMS), which is highly expressed in acute myeloid leukemia (AML) and associated with poor prognosis of AML, plays critical roles in leukemogenesis. Our study shows that inhibition of RBPMS inhibits self-renewal of leukemia-initiating cells (LICs) and leukemia development but has little effect on normal hematopoiesis. Mechanistically, RBPMS recruits the N6-methyladenosine (m6A) reader insulin-like growth factor 2 mRNA binding protein 3 (IGF2BP3), which promotes the stability of the forkhead box O1 (FOXO1) mRNA in an m6A-dependent manner. Moreover, RBPMS contributes to the progression of leukemia by directly binding to FOXO1 and promoting FOXO1-regulated glycolysis. Overexpression of FOXO1 has been shown to reverse RBPMS inhibition–induced phenotypes in both leukemic cells and mouse models. We also designed a specific inhibitor of RBPMS that has therapeutic effects in AML patient-derived xenograft (PDX) models. We therefore highlight RBPMS as a promising drug target for leukemia therapy.