2026-02-23 東京大学
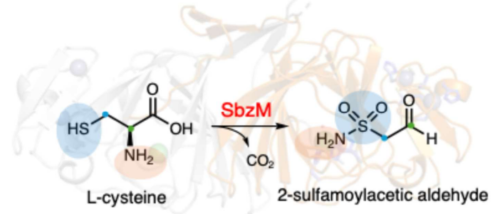
SbzM の構造と酵素反応
<関連情報>
- https://www.u-tokyo.ac.jp/focus/ja/press/z0111_00098.html
- https://www.u-tokyo.ac.jp/content/400280661.pdf
- https://www.nature.com/articles/s41929-026-01493-z
ニッケル依存性スルホンアミド合成酵素の構造・機能および機構解析 Structure–function and mechanistic analyses of nickel-dependent sulfonamide synthase
Yuhao Zhu,Takahiro Mori,Henrik P. H. Wong,Takayoshi Awakawa,Sam P. de Visser & Ikuro Abe
Nature Catalysis Published:23 February 2026
DOI:https://doi.org/10.1038/s41929-026-01493-z
Abstract
The sulfonamide group is an important functional group in biology and medicine that shows unique chemical properties and functions and is widely used in many marketed drugs. However, insertion of a sulfonamide group into a chemical scaffold is challenging and little is known about enzymatic sulfonamide biosynthesis. Here we show the structure, function and mechanism of the sulfonamide synthase SbzM, which is involved in the biosynthesis of altemicidin. Biochemical investigations established that SbzM strictly utilizes Ni2+ to convert L-cysteine into 2-sulfamoylacetic aldehyde via an initial decarboxylation followed by sulfur oxidation, representing a cysteine metabolism pathway distinct from those of canonical Fe2+-dependent cysteine dioxygenases. Further mechanistic studies, including site-directed mutagenesis, 18O-labelling, oxygen stoichiometry and computational studies, provided detailed insights into the SbzM-catalysed sulfonamide-formation reaction, which uses two dioxygen molecules and a Ni2+/Ni3+ redox cycle during catalysis. This study proposes a sulfonamide biosynthesis pathway with potential applications in biotechnology for sulfonamide synthesis through an environmentally benign process.

