2026-04-13 合肥物質科学研究院(HFIPS)
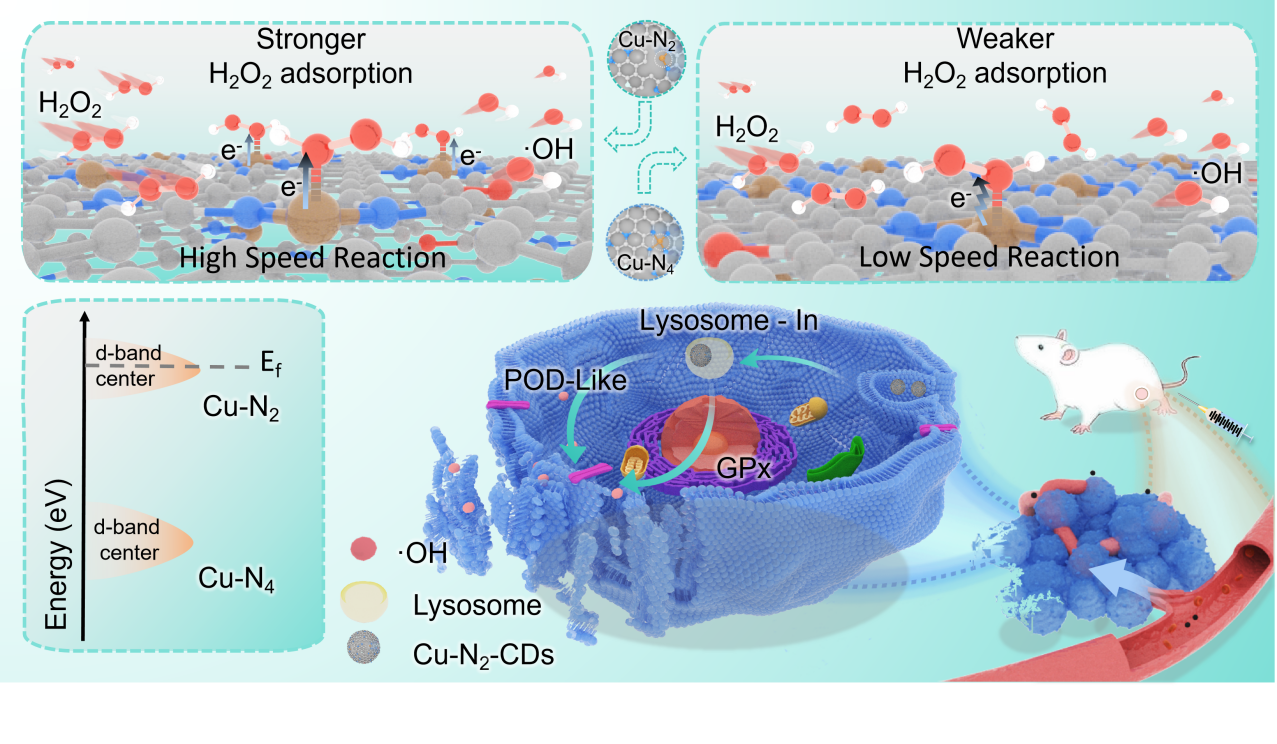
Schematic diagram of catalytic therapy mechanism of Cu-N2-CDs (Image by LIN Yefeng)
<関連情報>
- https://english.hf.cas.cn/nr/bth/202604/t20260413_1156089.html
- https://advanced.onlinelibrary.wiley.com/doi/10.1002/adfm.75092
炭素ドット上の配位不飽和Cu-N2単原子サイトを介した基質吸着と局所電子密度の調節による触媒療法の強化 Enhanced Catalytic Therapy by Modulating Substrate Adsorption and Local Electron Density through Coordinatively Unsaturated Cu-N2 Single-Atom Sites on Carbon Dots
Yefeng Lin, Chen Zhou, Yijun Lu, Yang Yang, Chenyang Bi, Xin Lai, Dongdong Wang, Junchao Qian, Songnan Qu, Hui Wang
Advanced Functional Materials Published: 25 March 2026
DOI:https://doi.org/10.1002/adfm.75092Digital Object Identifier (DOI)
ABSTRACT
Coordinatively unsaturated single-atom nanozymes with sufficient H2O2 adsorption and rapid kinetics display excellent peroxidase (POD)-like activity; however, their rational construction limits their wide application for cancer therapy. Here, we report a ligand chelation-determined conformation (LCDC) strategy to construct Cu single-atom nanozymes supported on carbon dots (Cu-N2-CDs) that were realized by in situ chelation and carbonization of organic chelating agent and metal cation. Such coordinatively unsaturated Cu-N2-CDs expose high-spin Cu sites that enhance d-p orbital hybridization to facilitate H2O2 adsorption. Importantly, the coordinatively unsaturated Cu-N2 configuration is capable of elevating the electron density at the metal center and narrowing the band gap to accelerate •OH generation reaction kinetics. The Cu-N2-CDs outperform their Cu-N4 counterparts by achieving a 3.49-fold stronger H2O2 adsorption and a 3.62-fold greater maximum reaction rate, leading to the efficient production of toxic •OH from endogenous H2O2. Consequently, Cu-N2-CDs achieve excellent tumor catalytic therapy outcomes both in vitro and in vivo. This work provides fundamental insights for the rational design of high-performance nanozymes through precise coordination engineering.