2026-04-13 合肥物質科学研究院(HFIPS)
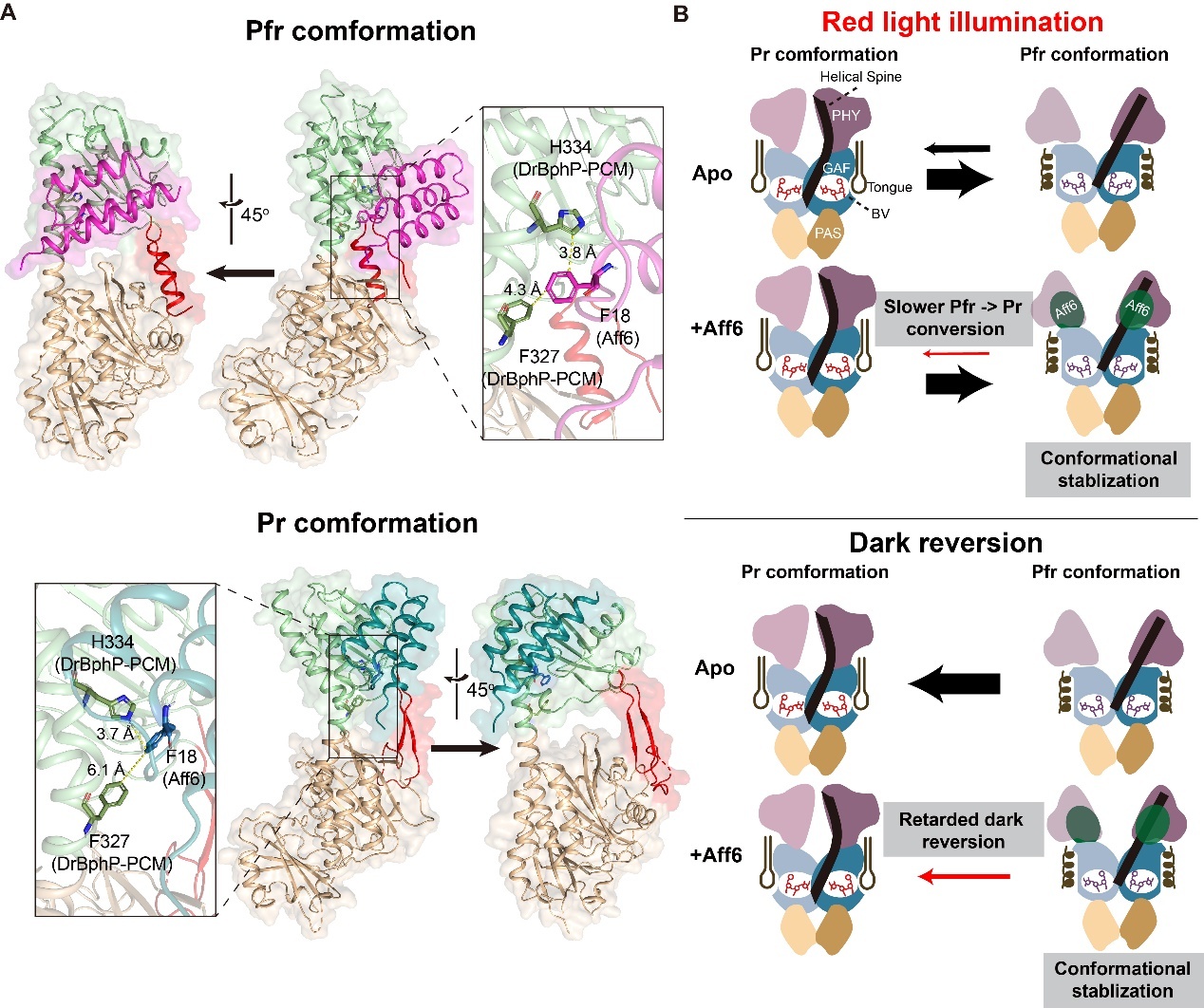
Molecular mechanism of the photo-state-dependent interaction between DrBphP and Aff6. (Image by ZHU Lei)
<関連情報>
- https://english.hf.cas.cn/nr/rn/202604/t20260413_1156108.html
- https://www.sciencedirect.com/science/article/abs/pii/S1011134426000783
アフィボディAff6の光状態特異的結合に関する構造的知見:DrBphPの光受容コアモジュールへの結合 Structural insights into photo-state-specific binding of affibody Aff6 to the photosensory core module of DrBphP
Lina Zhu, Runhan Wang, Mingyang Jin, Yingkui Dong, Hongxin Zhao, Junfeng Wang, Lei Zhu
Journal of Photochemistry and Photobiology B: Biology Available online: 25 March 2026
DOI:https://doi.org/10.1016/j.jphotobiol.2026.113431
Highlights
- The monomeric photosensory core module of DrBphP is sufficient to mediate light-inducible heterodimerization with Aff6.
- Aff6 primarily targets the PHY domain and the C-terminal region of the helical spine of DrBphP-PCMmono.
- Aff6 binding stabilizes the Pfr state of DrBphP-PCMmono and retards its Pfr-to-Pr dark reversion.
Abstract
Light-inducible heterodimerization systems offer precise, reversible control of protein interactions in living cells. Leveraging the high tissue-penetration of red/far-red light, the MagRed system, composed of a bacteriophytochrome Deinococcus radiodurans BphP (DrBphP) and its engineered affibody binder Aff6, achieves robust photoswitchable dimerization. This makes MagRed well-suited for in vivo and deep-tissue optogenetic application. However, the structural mechanism underlying Aff6’s photo-state-specific recognition of DrBphP remains elusive. Here, we combine solution NMR spectroscopy, surface plasmon resonance (SPR), molecular docking and mutational analysis to elucidate the light-dependent interaction between a monomeric photosensory core module of DrBphP (DrBphP-PCMmono) and Aff6. We show that DrBphP-PCMmono alone is sufficient for light-inducible heterodimerization with Aff6, exhibiting a ∼ 23-fold affinity difference between the Pfr and Pr states. NMR titration reveals that Aff6 binds primarily to the PHY domain and the C-terminal region of the helical spine. Furthermore, docking and mutagenesis identify a key aromatic interaction (involving F327/H334 of DrBphP and F18 of Aff6) as the molecular basis for this conformational selectivity. Additionally, Aff6 binding stabilizes the Pfr state and retards the Pfr-to-Pr reversion of DrBphP-PCMmono. These findings not only provide critical structural insight into MagRed function but also establish a foundation for rationally engineering next-generation phytochrome-based optogenetic tools.