2026-04-13 スイス連邦工科大学ローザンヌ校(EPFL)
<関連情報>
- https://actu.epfl.ch/news/when-oxygen-determines-if-a-limb-can-regrow/
- https://www.science.org/doi/10.1126/science.adw8526
種特異的な酸素感知が脊椎動物の四肢再生の開始を制御する Species-specific oxygen sensing governs the initiation of vertebrate limb regeneration
Georgios Tsissios, Marion Leleu, Kelly Hu, Alp Eren Demirtas, […] , and Can Aztekin
Science Published:9 Apr 2026
DOI:https://doi.org/10.1126/science.adw8526
Editor’s summary
Some vertebrates can regenerate limbs, whereas others cannot. By comparing regenerating frog tadpoles and nonregenerating mouse embryonic limbs, Tsissios et al. found that species-specific oxygen sensing determines whether amputation triggers limb regeneration (see the Perspective by Paoli and Whited). Frog tadpoles exhibited reduced oxygen sensing associated with diminished regulation of hypoxia-inducible factor 1A (HIF1A), enabling robust regeneration by promoting biomechanical, epigenetic, and metabolic states conducive to tissue regrowth. By contrast, mouse limbs displayed heightened sensitivity to oxygen, which destabilizes HIF1A and prevents regeneration. Lowering environmental oxygen levels or stabilizing HIF1A allowed mouse limbs to initiate regeneration. Mui et al. used a mouse digit amputation model to investigate why some injuries regenerate while others scar. They found that the extracellular matrix, the network of proteins and sugars surrounding cells, was crucial to regeneration. Regenerating tissue is soft, fluid, and rich in hyaluronic acid, whereas nonregenerating tissue is stiff and collagen heavy. Depleting hyaluronic acid halted regeneration and triggered scarring, whereas stabilizing it improved bone regrowth. —Stella M. Hurtley
Structured Abstract
INTRODUCTION
Some vertebrates, such as frog tadpoles and salamanders, can regenerate lost limbs after amputation, whereas mammals cannot. Many regeneration-associated molecular pathways and cellular programs are conserved across species, suggesting a possible latent limb-regenerative capacity for mammals. Nonetheless, it remains unclear why these pathways and cell types are not activated after limb amputation and whether limb regenerative programs can, in principle, be initiated in mammals.
RATIONALE
Direct functional comparisons of amputation responses across species are difficult in vivo because of physiological, environmental, and developmental differences, as well as practical constraints. We therefore used limb explants, tissues grown outside the body, as a highly controlled experimental platform. Having shown that frog tadpole (Xenopus laevis) limbs initiate regeneration as explants, we investigated whether embryonic mouse (Mus musculus) limbs do so under comparable conditions and, if not, which mechanisms distinguish regenerative from nonregenerative species.
RESULTS
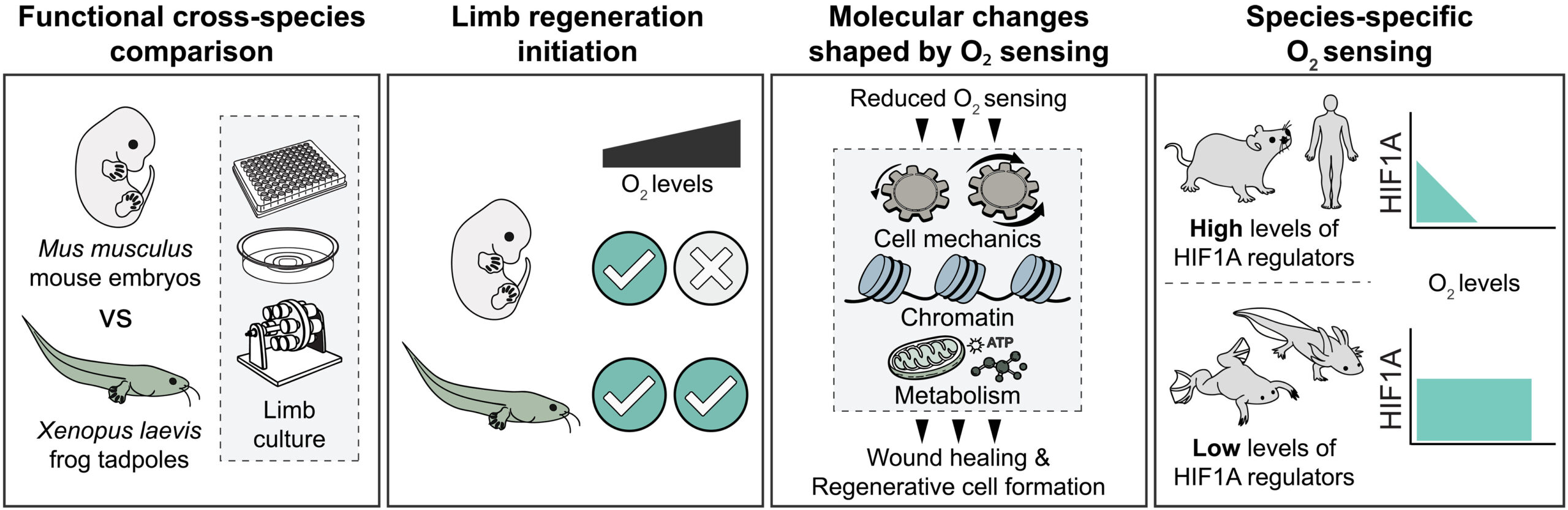
We found that subatmospheric oxygen conditions, or stabilization of the oxygen-sensitive transcription factor hypoxia-inducible factor 1A (HIF1A), promote rapid wound healing after amputation in embryonic mouse limbs. Reduced oxygen availability reshaped cellular biomechanical properties associated with YAP activation and metabolic states, particularly glycolysis. In parallel, it also rewired the chromatin landscape by decreasing the repressive histone mark H3K27me3 and increasing the activating mark H3K4me3, thereby permitting regenerative gene expression and the formation of limb regeneration–associated cell types. By contrast, atmospheric oxygen conditions impaired these processes in mouse limbs. In addition, frog tadpole limbs displayed robust wound healing, regenerative cell-type formation, and stable biomechanical, epigenetic, and metabolic features across a wide range of oxygen conditions, even those greatly exceeding atmospheric oxygen levels. This reduced oxygen sensing was associated with lower expression of HIF1A regulators, resulting in stable HIF1A activity relative to mice. Extending this analysis, we found that regenerative axolotls also show lower expression of HIF1A regulators, whereas humans exhibit a heightened oxygen-sensing signature similar to mice.
CONCLUSION
We propose species-specific oxygen-sensing capacity as a key determinant of limb regeneration initiation across vertebrates. Reduced oxygen sensing promotes biomechanical, epigenetic, and metabolic programs that are conducive to regeneration, with implications extending to development, disease, evolution, and cross-species comparisons. Finally, our findings demonstrate that modulation of oxygen-sensing pathways can unlock latent limb-regenerative programs in mammals, providing a mechanistic route toward inducing limb regeneration in adult mammals.
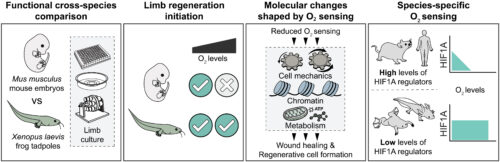
Species-specific oxygen sensing governs the initiation of vertebrate limb regeneration.
Cross-species comparison of frog tadpole and embryonic mouse limb explants reveals that low oxygen or reduced oxygen sensing enables the initiation of limb regeneration, coordinating biomechanical, chromatin, and metabolic states to promote wound healing and regenerative cell formation. Unlike mammals, amphibians exhibit reduced oxygen sensing, which is associated with sustained HIF1A activation across a wide range of environmental oxygen levels.
Abstract
Why mammals cannot regenerate limbs like amphibians do presents a long-standing puzzle in biology. To uncover the underlying differences, we compared amputation responses of embryonic mouse (Mus musculus) and Xenopus laevis tadpole limbs. Lowering environmental oxygen or stabilizing the oxygen-sensitive hypoxia-inducible factor 1A (HIF1A) induced rapid wound healing in mouse limbs. This response was accompanied by altered cellular mechanics, metabolism, and a histone landscape that primed regenerative cell states. Conversely, Xenopus tadpole limbs retained these features even under high oxygen levels. Their reduced oxygen-sensing capacity was associated with decreased HIF1A-regulating gene expression. Our results thus identify species-specific oxygen-sensing capacity as a fundamental, targetable mechanism that can unlock latent regenerative programs in mammals.