2026-04-14 関西学院大学
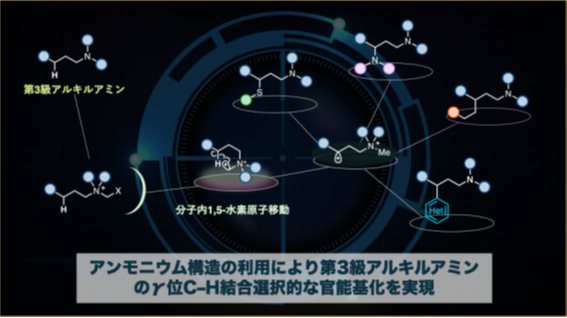
図1:本研究で開発した反応の概要。α-アンモニオラジカルを利用することで、第三級アルキルアミンのγ位C–H結合のみを選択的に官能基化することが可能になった。
<関連情報>
ジストニックラジカルカチオンを制御してアルキルアミンのγ-C–H官能基化を精密に行う
Taming distonic radical cations for precise γ-C–H functionalization of alkylamines
Takumi Kinoshita,Kazuki Hirate,Kosuke Hamawaki,Shoma Chiba,Kosuke Terada,Yota Sakakibara & Kei Murakami
Nature Synthesis Published:14 April 2026
DOI:https://doi.org/10.1038/s44160-026-01042-3
Alkylamines are crucial structural motifs found in 43% of active pharmaceutical ingredients, where they interact with biological targets. As a consequence, the C–H functionalization of alkylamines is a powerful strategy for accelerating drug development. Remote C–H functionalization enables selective modification of the periphery of the active nitrogen site and can improve bioactivity and pharmacokinetics. Unlike that of primary and secondary alkylamines, the remote functionalization of tertiary amines remains challenging, despite their prevalence in 60% of alkylamine-containing pharmaceuticals. This study exploits the reactivity of α-ammonio radicals, a class of distonic radical cations, to achieve the γ-selective functionalization of tertiary amines. Our approach uses halomethylammonium salts as α-ammonio radical precursors, facilitating precise radical transfer to the γ-position. The resulting γ-radicals enable diverse γ-selective C–H functionalizations, including thioetherification, amination, alkylation, (hetero)arylation and alkenylation. The developed method has a broad substrate scope and enables the late-stage functionalization of complex pharmaceutical molecules, thus holding promise for drug development. Furthermore, our work expands the synthetic use of distonic radical cations, broadening the methodological landscape for selective radical transformations and inspiring future advancements in radical chemistry.