2026-03-16 パシフィック・ノースウェスト国立研究所(PNNL)
<関連情報>
- https://www.pnnl.gov/news-media/protein-design-principles-revealed-ai-analysis-nanoribbon-assembly
- https://www.nature.com/articles/s41467-026-69170-0
溶媒力と対称性の破れが、液固界面における設計タンパク質の集合に及ぼす影響 Impact of solvent forces and broken symmetry on the assembly of designed proteins at a liquid-solid interface
Sakshi Yadav Schmid,Benjamin Helfrecht,Amy Stegmann,Benjamin A. Legg,Harley Pyles,Jiajun Chen,John R. Edison,Maxim Ziatdinov,Zdenek Preisler,Orion Dollar,Stephen Whitelam,Sergei Kalinin,David Baker,Christopher J. Mundy,Shuai Zhang & James J. De Yoreo
Nature Communications Published:13 March 2026
DOI:https://doi.org/10.1038/s41467-026-69170-0
Abstract
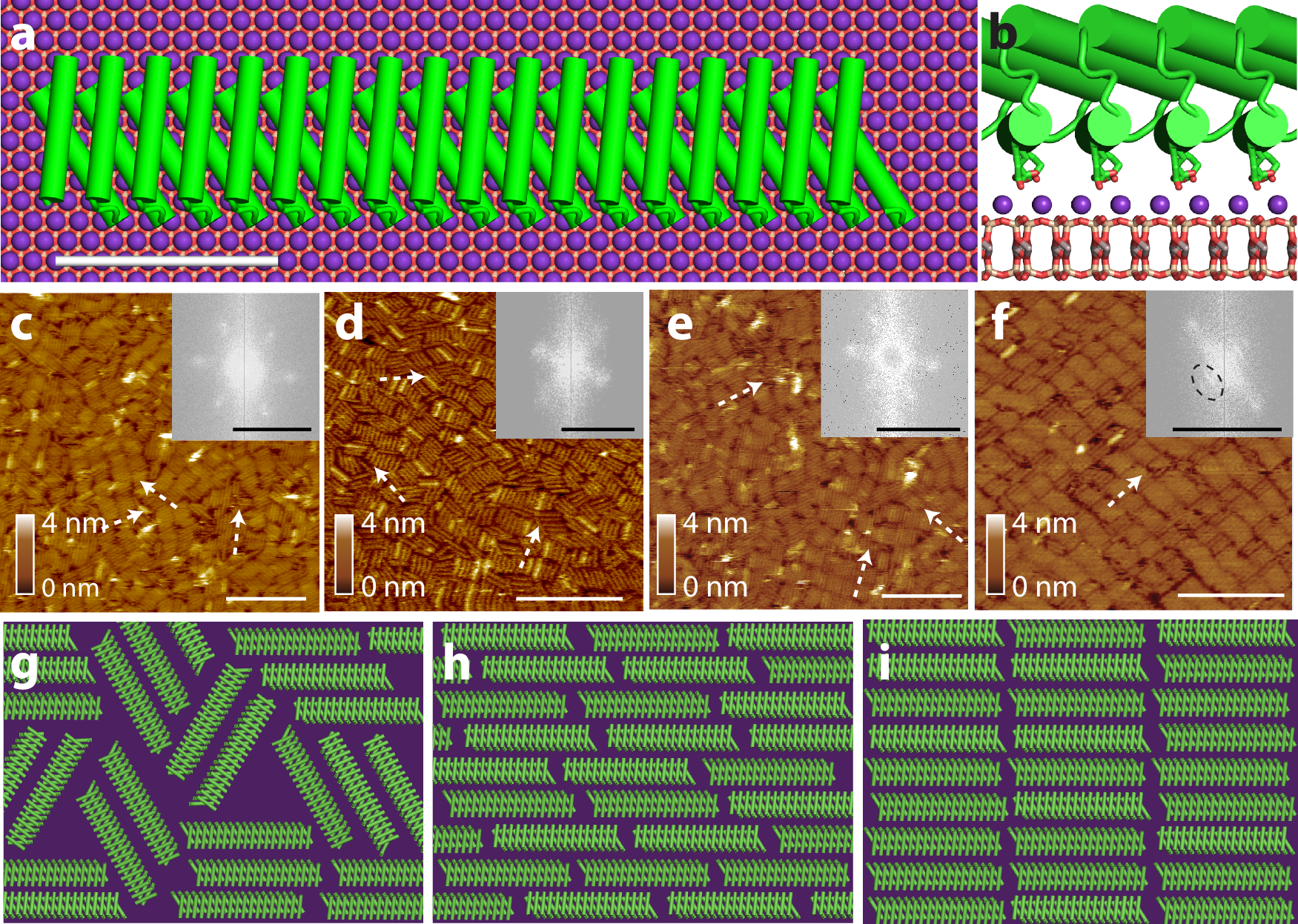
The era of protein design has enabled the creation of hybrid protein-inorganic interfaces, leading to both surface-directed self-assembly of de novo protein architectures and protein-directed formation of inorganic materials. However, the resulting patterns of protein assembly are often unexpected, implying that essential interactions are not accounted for in current design platforms. Here, we use high-speed atomic force microscopy (AFM) analyzed through machine learning to follow the assembly of protein nanorods in aqueous electrolytes on two types of mica exhibiting disparate symmetry elements, which are imprinted on the overlying hydration structure. Using Monte Carlo simulations, we reproduce the observed phases and show that an observed smectic phase, previously thought to be unstable for non-interacting rods in two dimensions, emerges when crystal symmetry introduces a directional bias. The findings demonstrate the importance of incorporating solvent forces as modulated by the hydration structure inherent to interfacial systems when designing protein assemblies at liquid-crystal interfaces. Coupling physics-based simulations that can account for these factors to de novo protein design algorithms can lead to improved design platforms for bio-inspired, hybrid materials.


