2026-04-07 東京大学

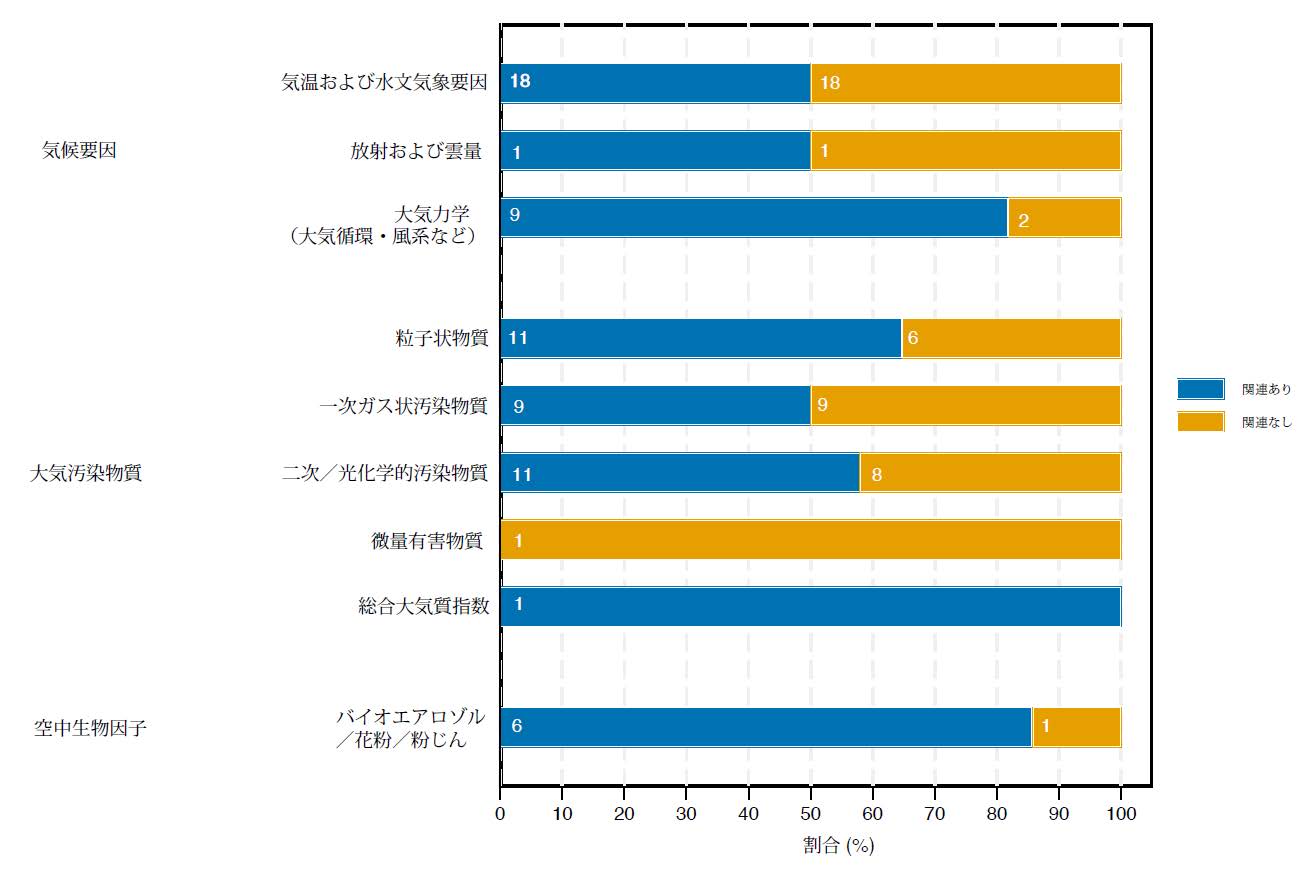
抗菌ペプチド(LL-37 と HNP1)のダブル協奏効果
上:ヒトの細胞膜上では抗菌ペプチドは集まり、「ブレーキモード」を示す
下:細菌の細胞膜上では抗菌ペプチドはほどけ、「攻撃モード」を示す
<関連情報>
- https://www.iis.u-tokyo.ac.jp/ja/news/5047/
- https://onlinelibrary.wiley.com/doi/10.1002/anie.202516436
凝集状態のダイナミクスが抗菌ペプチドLL-37とHNP1間の二重協同性を促進する Aggregation-State Dynamics Drive Double Cooperativity Between Antimicrobial Peptides LL-37 and HNP1
Yuge Hou, Jing Zhang, Koh Takeuchi, Hideo Higuchi, Motoshi Kaya, Rui Li, Jianlu Zheng, Jiali Chen, Go Watanabe, Kaori Sugihara
Angewandte Chemie International Edition Published: 02 April 2026
DOI:https://doi.org/10.1002/anie.202516436
ABSTRACT
The double cooperative effect between two major antimicrobial peptides, LL-37 and HNP1, where their combination enhances antimicrobial efficiency while reducing cytotoxicity, offers a promising strategy for developing safer and more effective antibiotics against antimicrobial resistance. However, the molecular mechanism driving this effect has remained unclear. In this study, a combination of total internal reflection fluorescence (TIRF) microscopy, Förster resonance energy transfer (FRET), nuclear magnetic resonance (NMR), and molecular dynamic simulation (MD) revealed that the double cooperative effect arises from the dynamic assembly and disassembly of LL-37/HNP1 aggregates, where the degree of aggregation governs membrane toxicity and therefore influences the cell fate. The aggregation is driven primarily through hydrophobic interactions between several amino acids within LL-37 and HNP1. Interestingly, in membranes, this aggregation state is regulated by the lipid composition, where only anionic lipids can break up the aggregates and disperse peptides in the membranes, leading to the gain of peptides’ function. Taking the advantage of the lipid composition difference, this enables the aggregates to destroy bacterial membranes, whereas protect eukaryotic cell membranes.