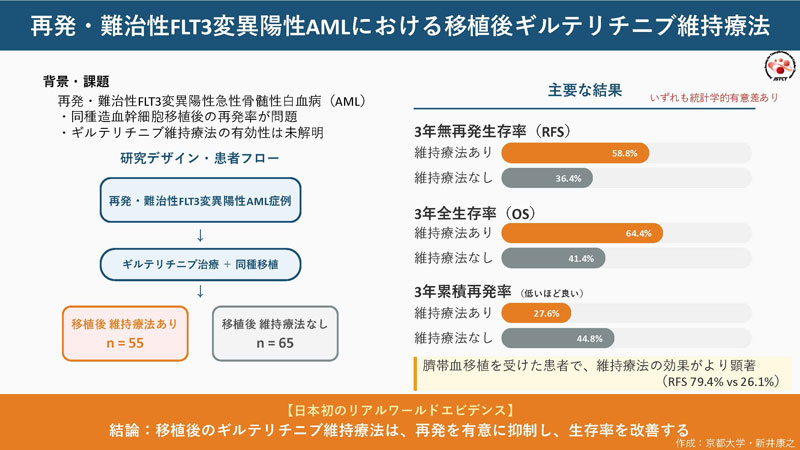
2026-04-17 北海道大学,東京科学大学
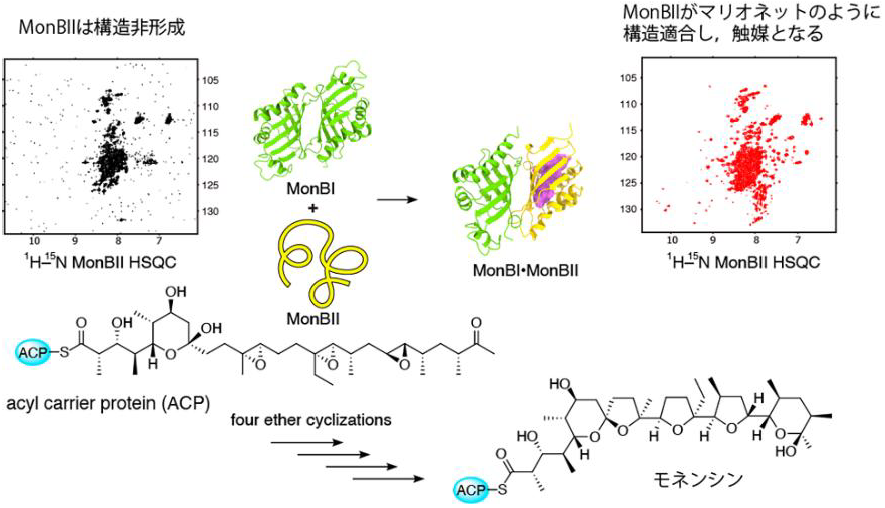
MonBIIは単独では構造を形成していないが、MonBIが存在すると、構造を形成する(証明は、HSQCという核磁気共鳴法の手法)。この構造形成が、モネンシンを作るための4回連続環化反応に必要であることが分かった。
<関連情報>
- https://www.hokudai.ac.jp/news/2026/04/post-2259.html
- https://www.hokudai.ac.jp/news/pdf/260417_pr.pdf
- https://www.nature.com/articles/s41557-026-02122-9
相方として鋳型をもちいた構造形成戦略により、ポリエーテル生合成連続環化反応が可能となる A system of paired polyether epoxide hydrolases enables a mouldable enzyme for consecutive ring cyclization cascades
Nana Yabuno,,Atsushi Minami,Taro Ozaki,Yuki Owada,Kohei Sawada,Ayaka Arai,Shusuke Sato,Aoi Sugiyama,Takashi Tadokoro,Tomoyasu Aizawa,Takuya Dosen,Tomo Nomai,Takanori Matsumaru,Junyang Liu,Tao Ye,Atsuji Kodama,Susumu Uchiyama,Kowit Hengphasatporn,Yasuteru Shigeta,Tomohide Saio,Katsumi Maenaka,Min Yao,Hiroyuki Kumeta,Hideaki Oikawa &Toyoyuki Ose
Nature Chemistry Published:16 April 2026
DOI:https://doi.org/10.1038/s41557-026-02122-9
Abstract
Ionophore polyethers, a major class of polyketide-derived natural products, are characterized by molecular skeletons featuring arrays of tetrahydrofuran and tetrahydropyran rings. However, the precise mechanism underlying their biosynthesis, suggested to occur by a sequential epoxide-opening and ether cyclization cascade to generate more than two ether rings, remains elusive. Here we explore the biosynthesis of monensin and reveal the indispensability of a heterodimeric assembly of polyether epoxide hydrolases (MonBI·MonBII), with MonBII providing the sole active site. Structural analysis demonstrated that MonBII harbours an unusually large cavity, enabling the consecutive cyclization of substrates containing γ-hydroxy triepoxide moieties. This cavity is formed by the remarkable flexibility of MonBII, which undergoes a dramatic structural transition from a predominantly disordered state to its active folded conformation exclusively in the presence of MonBI. Given the widespread conservation of MonBI- and MonBII-type enzymes, this study emphasizes a unified pairing mechanism, wherein one protomer functions as a molecular mould, facilitating the folding process and stabilizing the structure of its partner for catalysis.