2026-05-01 京都大学
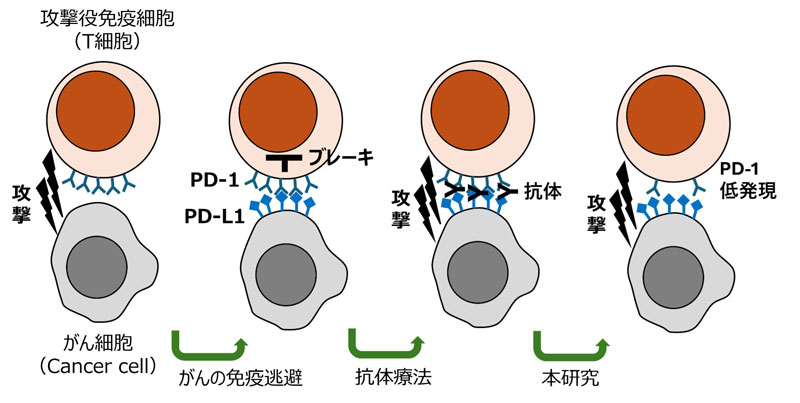
従来の抗体療法は外からブレーキを阻害するが、本研究は遺伝子操作でPD-1を低発現に保ち、がん細胞の免疫逃避を防いで攻撃力を高める新しいアプローチへの基礎研究である。(図作成:SEO Wooseok)
<関連情報>
- https://www.kyoto-u.ac.jp/ja/research-news/2026-05-01-2
- https://www.sciencedirect.com/science/article/pii/S0165247826000441
Pdcd-1 上流エンハンサーの遺伝子欠損はT細胞機能と抗腫瘍応答を強化する Genetic disruption of Pdcd-1 upstream enhancer boosts T cell function and antitumor response
Chandsultana Jerin, Wooseok Seo, Hiroyoshi Nishikawa
Immunology Retters Available online: 27 March 2026
DOI:https://doi.org/10.1016/j.imlet.2026.107171
Highlights
- Established an upstream enhancer knockout (UpEnh KO) strain using CRISPR–Cas9 genome editing to dissect Pdcd-1 regulation in T cells.
- UpEnh deletion reduced PD-1 expression across multiple T cell subsets, including thymocytes, peripheral naïve Treg, and γδ T cells, as well as tumor-infiltrating exhausted CD8+, CD4+ Tconv, Treg, and γδ T cells.
- Loss of UpEnh from the Pdcd-1 locus improved CD8+ T cell exhaustion state, boosted γδ T cell activity, and promoted more robust anti-tumor responses.
Abstract
Programmed cell death 1 (PD-1) is an inhibitory receptor that drives T cell exhaustion in tumors, limiting antitumor immunity. Current PD-1 blockade therapies have shown limited success. To uncover new strategies for modulating PD-1, we investigated an upstream enhancer (UpEnh) of the Pdcd-1 gene using a CRISPR-Cas9 knockout mouse model. Deletion of the UpEnh reduced PD-1 expression across various T cell subsets. In a tumor setting, this deletion lowered PD-1 levels on intratumoral exhausted CD8⁺, conventional CD4⁺, Treg, and γδ T cells. This resulted in improved CD8⁺ and γδ T cell function and promoted stronger antitumor immunity. Our findings establish UpEnh as a critical regulator of PD-1, presenting a potential therapeutic target.