2026-05-11 国立精神・神経医療研究センター
<関連情報>
- https://www.ncnp.go.jp/topics/detail.php?@uid=57hXKUTs1zU02en1
- https://www.nature.com/articles/s41380-026-03559-7
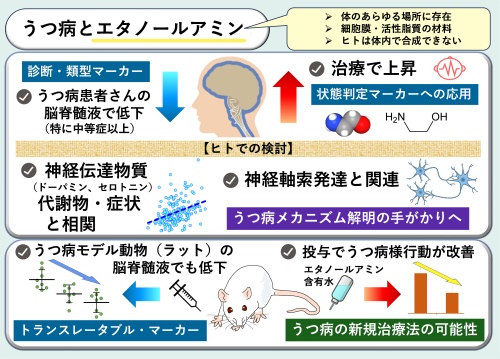
エタノールアミンはうつ病の潜在的なバイオマーカーおよび治療標的となる可能性がある Ethanolamine as a potential biomarker and therapeutic target for depressive disorder
Shintaro Ogawa,,Kotaro Hattori,Shinsuke Hidese,Daimei Sasayama,Miho Ota,Tomoko Miyakawa,Megumi Tatsumi,Ryo Matsumura,Sumiko Yoshida,Takamasa Noda,Minoru Takebayashi,Wataru Omori,Kei Itagaki,Naoto Kajitani,Mami Okada-Tsuchioka,Akitoyo Hishimoto,Shuken Boku,Tadasu Horai,Akira Monji,Yoshito Mizoguchi,Hiroshi Tateishi,Toru Murakawa-Hirachi,Reiji Yoshimura,Ryohei Igata,… Hiroshi Kunugi
Molecular Psychiatry Published:15 April 2026
DOI:https://doi.org/10.1038/s41380-026-03559-7
Abstract
We investigated cerebrospinal fluid (CSF) ethanolamine (EA) levels in major depressive disorder (MDD), to validate our previous findings on EA (Ogawa et al., 2015) and broaden its biological context and translational relevance. Using human samples (sets A–C) and animal models, we explored the implications for replication, translational potential, therapeutic development, and mechanistic insight. In set A (n = 380), CSF EA levels were low in patients with MDD (P = 0.00047, Cohen’s d = −0.59), negatively correlating with depression severity (Spearman’s ρ = −0.29, P = 0.00015) and positively correlating with CSF homovanillic acid (partial r = 0.40, P = 6.7E − 8) and 5-hydroxyindoleacetic acid levels (partial r = 0.26, P = 0.00064). Patients with moderate-to-severe depression showed large effects regardless of medication (Cohen’s d = −1.27 to −1.14). In set B (n = 13), CSF EA levels were significantly increased in patients who received electroconvulsive therapy (P = 0.0071; Cohen’s d = 0.90), which was linked to Hamilton Depression Rating Scale subscale score recovery. In set C (n = 66), a novel multicenter-collected sample set, CSF EA levels were low in patients with MDD (P = 0.0037; Cohen’s d = −0.91). Rats receiving 0.5 or 1.0 mg/kg lipopolysaccharide intraperitoneally for 7 days showed significantly reduced CSF EA levels (P = 0.014; Cohen’s d = −1.52 and 0.00020; d = 2.31, respectively) and depressive-, anxiety-, and anhedonia-like behaviors. Rats orally administered EA for 4 weeks showed significant antidepressant-like behaviors (P = 0.016; Cohen’s d = −1.36). Proteomic and bioinformatic analyses revealed 40 proteins that were significantly correlated with CSF EA levels. The top hit protein was CHL1 (partial r = 0.42, P = 1.5E − 9), and the axon guidance pathway was the most enriched (P = 3.4E − 20). EA is a potential CSF biomarker for mental state evaluation, treatment response, subtyping, and validation of MDD animal models and a promising research tool for developing a new classification framework for psychiatric disorders and novel therapeutic strategies.